Neovascular glaucoma (NVG) surgery is usually the lastline treatment that brings eye pressure down when medicines and laser cant stop it. Its the option that doctors turn to when the disease threatens your vision despite all other efforts.
Why does that matter? Because the right surgery can preserve the sight you still have, but every operation carries benefits, risks, and a recovery curve. Below youll get the full picture, so you can walk into the appointment confident, informed, and a little less anxious.
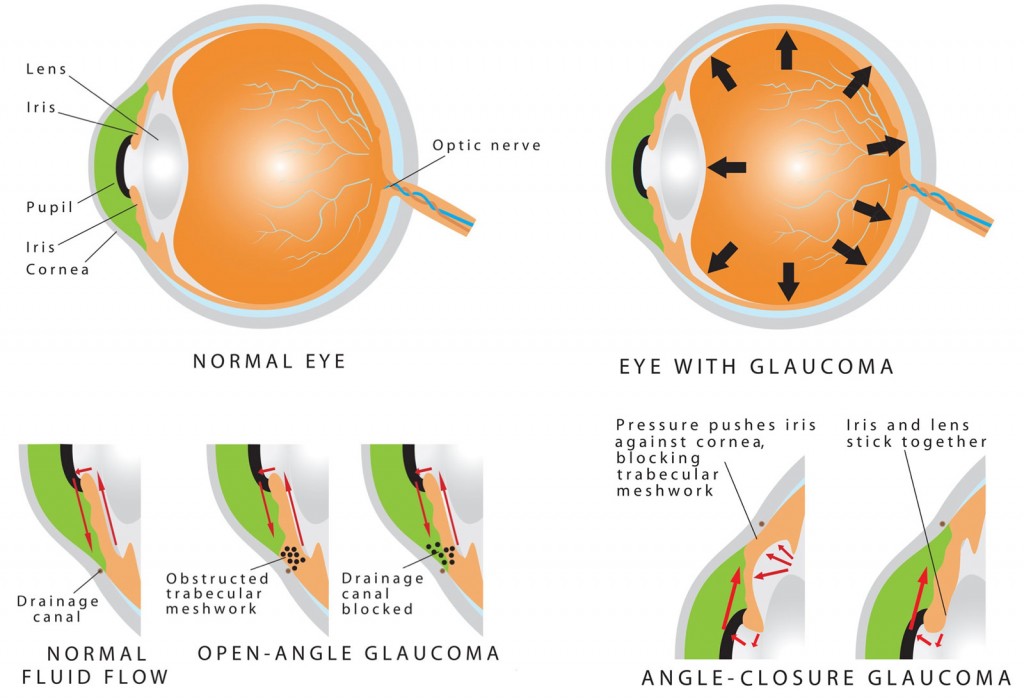
When Surgery Needed
What are the stages of neovascular glaucoma?
NVG doesnt appear overnight. It usually progresses through three recognizable stages, each with its own clues:
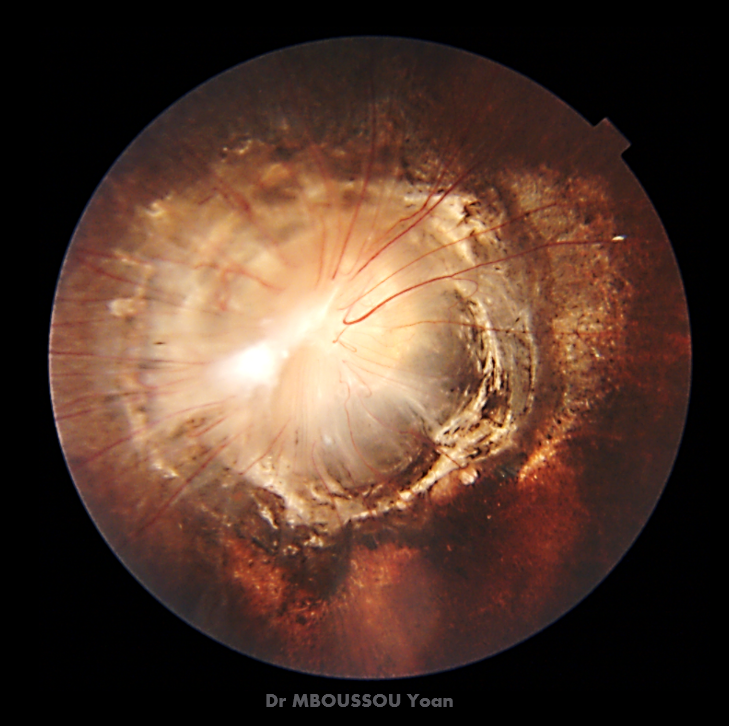
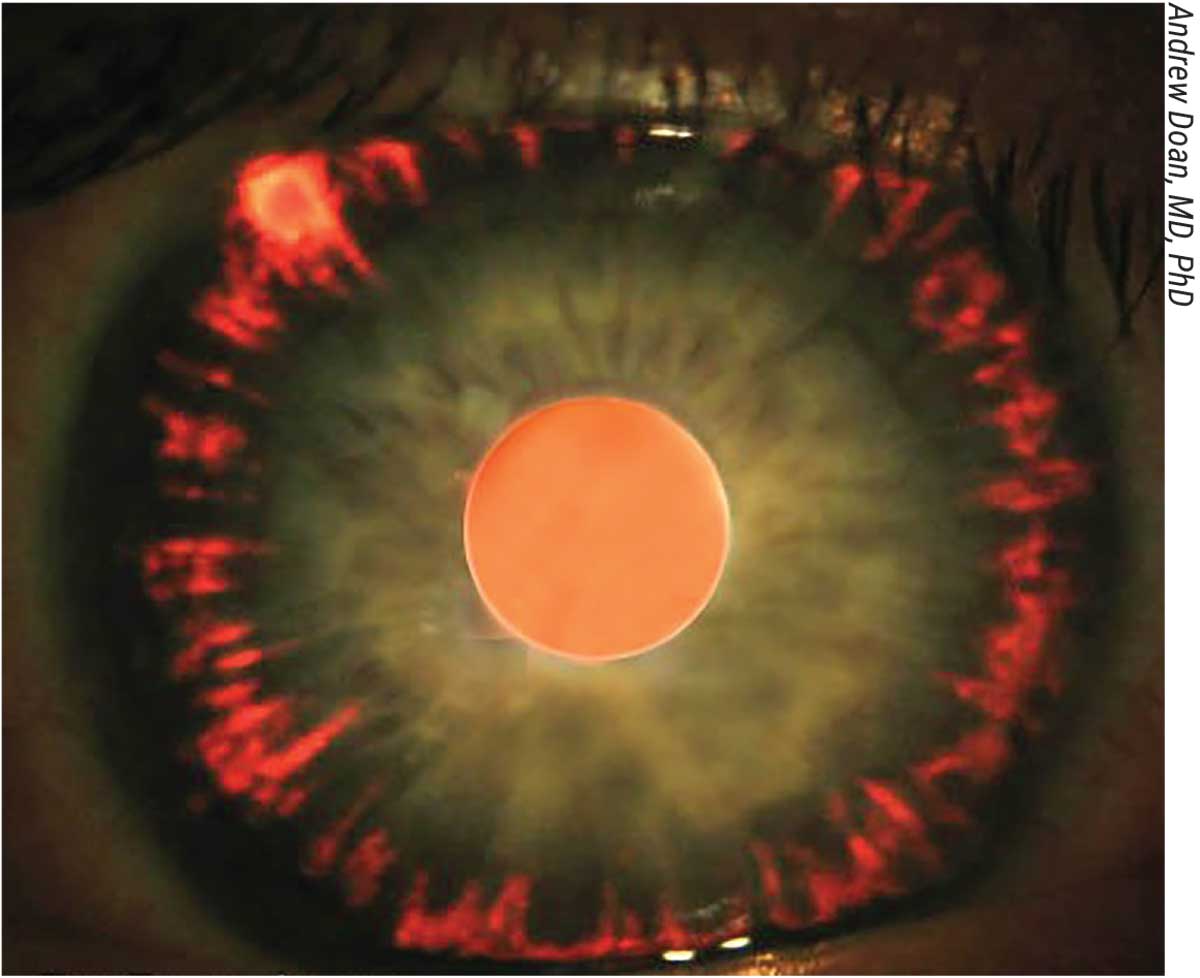
- Stage1 early neovascularisation. Tiny new vessels appear on the iris (rubeosis) and in the drainage angle. At this point, most patients still respond to and laser.
- Stage2 progressive angle involvement. Vessels grow thicker, the angle closes, and intraocular pressure (IOP) starts to rise steadily.
- Stage3 refractory pressure. IOP stays high despite maximal medication. Vision may already be compromised, and surgery becomes the most realistic chance to protect whats left.
What pushes NVG toward surgery?
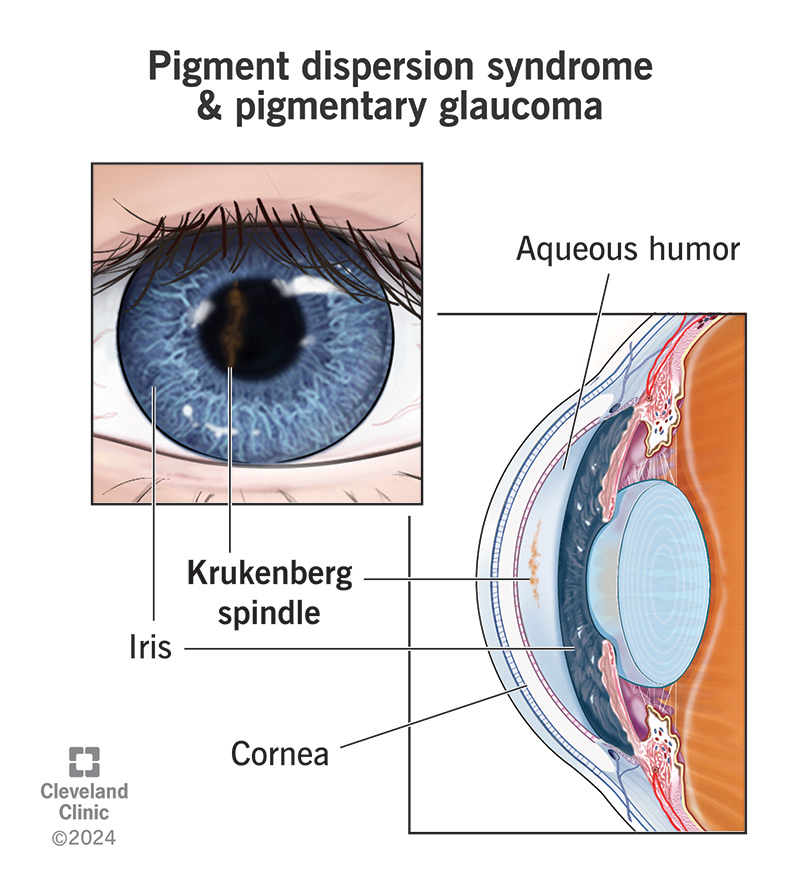
The underlying culprits are usually diseases that starve the retina of oxygen, prompting it to release VEGF (vascular endothelial growth factor). Common include:
- Diabetic retinopathy
- Central retinal vein occlusion (CRVO)
- Ocular ischemic syndrome
In my own clinic, I once saw a 58yearold teacher who had battled diabetic eye disease for years. Her eyes suddenly flooded with new vessels, her pressure shot up, and despite aggressive drops, she still hovered at 38mmHg. Thats the exact moment we started talking about surgery.
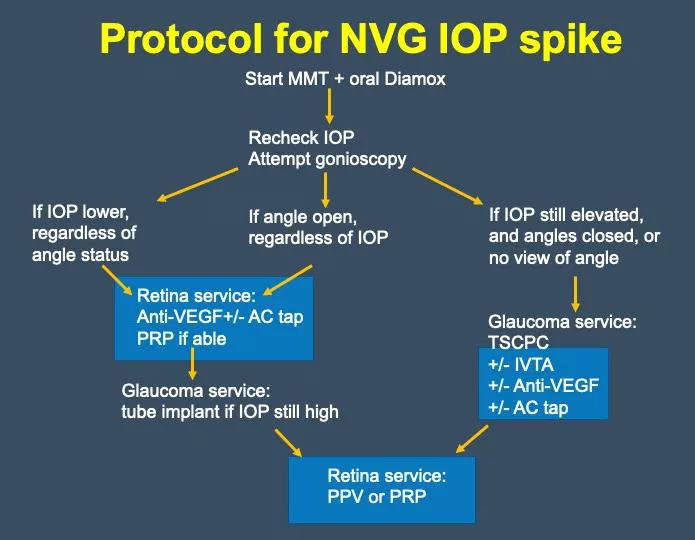
Surgical Options
What procedures are on the table?
There isnt a onesizefitsall surgery for NVG. Heres a quick tour of the most common techniques, each with its own sweet spot:
| Procedure | When It Shines | Main Advantage | Typical Risk |
|---|---|---|---|
| Glaucoma drainage implant (tube shunt) | Stage3, poor visual potential | Consistently lowers IOP | Tube erosion, hypotony |
| Trabeculectomy + antimetabolite | Earlymid stages, good visual potential | Less hardware in eye | Bleeding, scarring |
| Cyclophotocoagulation (laser) | Very low vision, endstage | Minimally invasive | Transient inflammation |
| Pars plana vitrectomy + antiVEGF | Concurrent retinal disease | Treats retina + glaucoma together | Longer surgery, cataract risk |
How do doctors decide?
Think of the choice like picking a pair of shoes: you consider the terrain, the fit, and the style. The decisionmaking checklist usually includes:
- How far the disease has progressed (stage)
- Whats causing the neovascularisation (e.g., diabetes, CRVO)
- How much useful vision remains
- Patients general health and ability to tolerate anesthesia
- Surgeons experience with each technique
When I discuss options with patients, I always bring a printed that visually breaks down each method. Seeing the pros and cons sidebyside makes the conversation feel less intimidating.
Inside The OR
What should you expect before the knife?
First, the ophthalmologist will run a full eye exam, take IOP measurements, and likely order retinal imaging (OCT or fluorescein angiography). Most surgeons also give an antiVEGF injection a week or two ahead of time it calms the new vessels and reduces bleeding risk during the operation.
Stepbystep of a typical tubeshunt implant
- Anesthesia. Usually a local block with a mild sedative; some patients prefer general anesthesia.
- Conjunctival peritomy. The surgeon lifts the thin membrane covering the eye to expose the sclera.
- Plate positioning. A silicone plate is tucked behind the eye muscles, often in the superior quadrant.
- Tube insertion. A tiny tube is threaded into the anterior chamber, allowing fluid to drain onto the plate.
- Securing and closing. Sutures hold everything in place, and the conjunctiva is closed with fine stitches.
Throughout the procedure, a small amount of bleeding may occur. Thats why the preop antiVEGF dose is so helpful its like sending a quietplease note to the new vessels.
Recovery timeline
Postop care is a marathon, not a sprint:
- First 24hours: Steroid drops to tame inflammation; IOP check before you go home.
- Week1: Another IOP check, possible adjustment of drops.
- Month1: Look for the hypertensive phase, a temporary rise in pressure that happens in up to 30% of patients. It usually settles with medication.
- Longterm (36months): Most surgeons aim for IOP <21mmHg without additional meds. Ongoing retinal laser (panretinal photocoagulation) may still be needed.
Pros vs Cons
What are the upside benefits?
When the surgery goes well, youll notice a rapid drop in pressure, often within hours. That can:
- Preserve any remaining peripheral vision
- Reduce or eliminate the need for multiple daily eye drops
- Lower the risk of painful optic nerve damage
Whats the flip side?
No medical decision is riskfree. Common complications include:
- Early hypotony (pressure too low) or postoperative hypertension
- Tube exposure or erosion, which may require a revision surgery
- Bleeding, infection, or cataract formation (especially with pars plana approaches)
- Transient double vision (diplopia) due to extraocular muscle irritation
Experienced surgeons mitigate many of these risks by using antimetabolites, meticulously placing the tube away from the cornea, and scheduling tight followup appointments.
Adjunct Treatments
How do antiVEGF drugs fit in?
Agents like bevacizumab, ranibizumab, and aflibercept work by blocking the VEGF signal that fuels neovascular growth. A typical regimen before surgery is a single injection 12weeks ahead, followed by another at the time of the operation if needed. According to a 2023 study in Frontiers in Ophthalmology, this strategy cuts intraoperative bleeding by up to 40%.
Why is panretinal photocoagulation still essential?
Even after the pressure is under control, the underlying retinal ischemia remains. Panretinal photocoagulation (PRP) burns peripheral retinal tissue to reduce its oxygen demand, thereby lowering VEGF production. Without PRP, the new vessels can reform, and the pressure may creep back up.
Can neovascular glaucoma be cured?
In short, the disease is rarely cured in the absolute sense. The goal is longterm control. When you combine surgery, antiVEGF therapy, and PRP, the odds of stable IOP and preserved vision improve dramatically. Think of it as a team sport rather than a solo act.
Success Data
What do recent numbers tell us?
Hospitalbased outcomes from 20222024 show:
- Tube shunts achieve target IOP (<21mmHg) without meds in about 65% of eyes.
- Trabeculectomy, when augmented with mitomycinC, reaches similar success rates but carries a higher risk of early blebrelated infections.
- Cyclophotocoagulation is effective for eyes with <5/200 vision, providing a pressure drop in roughly 55% of cases.
Patientreported qualityoflife surveys consistently note relief from the nightly pressure spikes that once disrupted sleep. One 60yearold patient summed it up: I can finally read my grandchildrens letters without fearing my eyes will shut down.
Your Action Plan
What should you check off before booking surgery?
- Confirm a full retinal workup, including OCT and fluorescein angiography.
- Schedule an antiVEGF injection at least one week before the procedure.
- Ask your eyedoctor about their experience with NVG how many tube shunts have they placed in the past year?
- Discuss both tube shunt and trabeculectomy options, and request a prosandcons list tailored to your eyes condition.
- Arrange postop support: someone to drive you home, help with eyedrop schedules, and watch for red flags (pain, sudden vision loss, severe redness).
How to prepare at home?
Keep a medication list handy, note any allergies, and write down questions you want to ask. Simple things like Will I need a protective shield after surgery? or When can I return to normal activities? can make the day feel less overwhelming.
Conclusion
Neovascular glaucoma surgery is a powerful, yet nuanced, option for halting the pressure spikes that threaten sight. By understanding the disease stages, weighing the surgical toolbox, and embracing the supporting roles of antiVEGF drugs and laser photocoagulation, you can approach the decision with confidence. Talk to a boardcertified glaucomaretina specialist, bring your checklist, and remember that youre not alone many have walked this path and emerged with clearer vision and peace of mind. If you have questions, share your thoughts in the comments, or reach out to a trusted eyecare professional. Your eyes deserve the best care, and you deserve to feel empowered every step of the way.
FAQs
When is neovascular glaucoma surgery recommended?
It is considered when intra‑ocular pressure stays high despite maximal drops and laser, usually in stage 3 NVG where vision is at risk.
What are the main types of surgery for neovascular glaucoma?
Common procedures include glaucoma drainage implants (tube shunts), trabeculectomy with antimetabolites, cyclophotocoagulation, and combined pars plana vitrectomy with anti‑VEGF therapy.
How long is the recovery period after a tube‑shunt implant?
Patients typically use steroid drops for the first 24 hours, have follow‑up checks at 1 week and 1 month, and may see a “hypertensive phase” that resolves with medication within a few weeks.
What role do anti‑VEGF injections play before surgery?
Giving an anti‑VEGF agent 1–2 weeks prior reduces neovascular bleeding during the operation and helps improve postoperative outcomes.
What are the common risks and complications of NVG surgery?
Potential issues include early hypotony or postoperative hypertension, tube exposure, infection, cataract formation, and transient double vision.