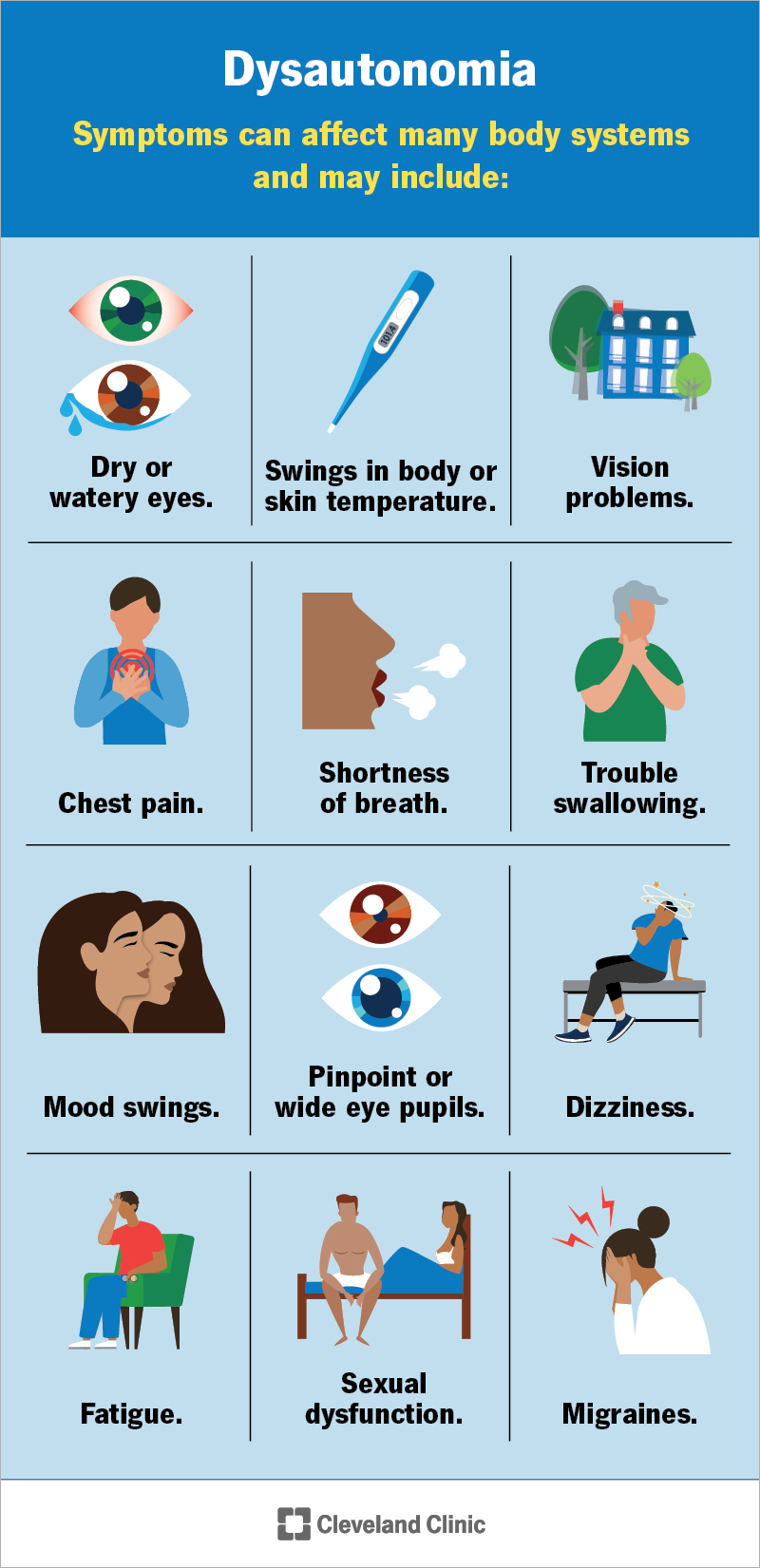
If you or someone you love has Duchenne muscular dystrophy (DMD) with an exon51amenable mutation, youve probably heard the name Amondys51 tossed around a lot lately. In plain terms, Amondys51 (casimersen) is the FDAapproved medicine that helps the body skip faulty exon51 so a shorter, but still functional, dystrophin protein can be made. Below, Ill walk you through exactly how it works, who can take it, the realworld ups and downs, and what you need to know before you start. Think of this as a friendly chat over coffeenot a dense textbook.
What Is Amondys51?
Definition & Brand Name
Amondys51, marketed as casimersen, belongs to a class of medicines called antisense oligonucleotides. These tiny strands are designed to bind to a specific piece of genetic codeexon51in the dystrophin gene. By doing so, they tell the cell to skip that exon during protein production, allowing a truncated yet functional dystrophin to be formed.
How It Differs From Exondys51
Many people mix up Amondys51 with Exondys51 (eteplirsen). While both target exon51, they are completely different molecules with distinct dosing schedules and FDA labeling. Below is a quicklook table that highlights the main differences.
| Drug | Active Ingredient | Mechanism | FDA Approval Year | Target Exon |
|---|---|---|---|---|
| Amondys51 | Casimersen | Antisense oligonucleotide exon51 skipping | 2021 | 51 |
| Exondys51 | Eteplirsen | Phosphorodiamidate morpholino oligomer exon51 skipping | 2016 | 51 |
How Exon Skipping Works
The Science in Plain Language
Imagine your DNA is a book and each exon is a page. In some DMD patients, the page labeled 51 is badly torn, making the whole story unreadable. Exonskipping therapy hands the cell a tiny bookmark (the antisense oligo) that tells it to simply leave that torn page out and keep reading the rest. The result? A shorter, but still coherent, narrativei.e., a dystrophin protein that can still do a decent job holding muscle cells together.
Antisense Oligonucleotides How They Do Their Job
These molecules slide into the cells nucleus and bind to the premRNA exactly where exon51 sits. The cellular splicing machinery then removes that exon before the mRNA is turned into protein. The process is called exon51 skipping. You can read more about the FDAs approval details Exondys 51 assistance.
Who Can Use It?
Genetic Mutation Requirements
To be eligible, a patient must have a confirmed DMD mutation that is amenable to exon51 skipping. This includes deletions, duplications, or point mutations that involve exon51 or its neighboring exons (for instance, a dmd exon 4951 deletion or a dmd exon 5152 deletion). Genetic testing from a certified lab is the only reliable way to verify this.
Age and Clinical Criteria
The FDA label states that Amondys51 is approved for patients aged 2years and older who are still ambulatory (i.e., able to walk). The drug is administered via weekly intravenous infusion, so families need to have access to a qualified infusion center.
How to Verify Eligibility A Simple Checklist
| Step | What You Need |
|---|---|
| 1. Genetic Test | Report showing exon51amenable mutation (e.g., dmd exon 4951 deletion) |
| 2. Clinical Evaluation | Baseline cardiac and respiratory assessments, ambulatory status check |
| 3. Insurance Review | Priorauthorization forms from Medicare/Medicaid or private insurer |
Benefits and Outcomes
What the Trials Showed
The pivotal 48week openlabel study (see the ) demonstrated that patients receiving Amondys51 showed a modest but statistically significant improvement in the sixminute walk test (6MWT) compared with historical controls. In plain speak, many boys were able to walk farther in six minutes after a year of treatment.
RealWorld Stories
Take Liam, a 7yearold who started Amondys51 right after his genetic report confirmed an exon4951 deletion. Within six months, his parents noticed he could climb stairs with less fatigue and even joined a soccer clinic for the first time in years. While not every child experiences the same boost, these anecdotes underline the genuine hope the drug can bring.
When Benefits Are Most Noticeable
Early initiationideally before significant loss of ambulationtends to give the best chance for preserving muscle function. Thats why pediatric neurologists stress getting the genetic test as soon as DMD is suspected.
Risks and Side Effects
Common Side Effects
Most patients report mild infusionrelated reactions such as headache, nausea, or a brief fever. Lab monitoring often shows mild elevations in serum creatinine, reflecting the drugs renal clearance. These changes are usually reversible after a dose adjustment.
Rare but Serious Events
In rare cases, patients have experienced hypersensitivity reactions, including rash or shortness of breath. Because the drug is filtered through the kidneys, severe renal impairment is a contraindication. Ongoing monitoring of kidney function before each infusion is crucial.
Monitoring Recommendations
| Test | Frequency | Threshold for Action |
|---|---|---|
| Serum Creatinine | Every infusion | Increase >0.5mg/dL from baseline discuss dose hold |
| Liver Enzymes (ALT/AST) | Monthly | >3 ULN consult physician |
| Cardiac Echo | Every 6months | Any decline in ejection fraction adjust care plan |
Comparing Other ExonSkipping Drugs
Amondys45 (Casimersen) Exon45 Skipping
Amondys45 targets a different mutation (exon45). Its approved for patients with deletions amenable to exon45 skipping, and the dosing schedule is identicalweekly IV infusion.
Vyondys53 (Golodirsen) Exon53 Skipping
Vyondys53 works on yet another slice of the DMD genetic puzzle. While its safety profile is similar, the efficacy data are still emerging, and the drug is priced comparably to Amondys51.
Key Comparison Chart
| Drug | Target Exon | Age Range | Typical Efficacy (6MWT) | Annual Cost (USD) |
|---|---|---|---|---|
| Amondys51 | 51 | 2years | +30min 6MWT (average) | $300,000 |
| Amondys45 | 45 | 2years | +20min 6MWT (average) | $300,000 |
| Vyondys53 | 53 | 4years | Data limited | $300,000 |
Regulatory Status & Insurance Considerations
FDA Approval Timeline
Amondys51 received its full FDA approval in March2021 after an accelerated review based on promising earlyphase data. The drugs label includes specific dosing instructions, safety monitoring guidelines, and the requirement that patients be ambulatory.
PriorAuthorization Steps
Most insurersboth public (Medicare, Medicaid) and privaterequire a priorauthorization packet that includes the genetic report, a letter of medical necessity, and a dosing schedule. The manufacturer provides a downloadable PDF that guides families through this process. For help navigating coverage and patient support options, see Exondys 51 insurance.
Cost & Assistance Programs
At roughly $300,000 per year, the price is steep. However, the sponsor offers a copayassistance program, and many nonprofits provide grant funding for families who meet income criteria. Its worth calling the patientsupport hotline early to explore these options.
RealWorld Voices: Patient & Caregiver Stories
A Short Narrative
When Mayas 5yearold son, Ethan, was diagnosed with a dmd exon5152 deletion, the family faced a whirlwind of emotionsfear, hope, and a lot of paperwork. After confirming eligibility, they started weekly infusions at a local children\'s hospital. Six months later, Ethan began to regain some of the confidence hed lost on the playground. Its not a miracle cure, Maya says, but it gave us a fighting chance and a reason to celebrate the small victories.
Caregiver Checklist for Home Prep
- Set up a dedicated infusion space (quiet, comfortable chair).
- Keep a medication diary to track any side effects.
- Maintain a calendar for lab draws and doctor appointments.
- Connect with a local DMD support group for emotional backup.
TakeAway Checklist Before Starting Treatment
Eligibility Verification
- Obtain a certified genetic test confirming exon51 amenability.
- Document ambulatory status and baseline cardiac/respiratory function.
- Gather insurance priorauth forms and manufacturer assistance paperwork.
Safety & Monitoring Plan
- Schedule weekly infusion appointments with a qualified IV team.
- Arrange labs (creatinine, liver enzymes) before each dose.
- Plan a 6month cardiac echo followup.
Financial & Insurance Prep
- Contact the drugs patientsupport line for copay help.
- Submit the priorauthorization packet early to avoid delays.
- Explore local charitable grants for raredisease therapies.
Having all these pieces in place turns a potentially overwhelming process into a manageable roadmap.
Conclusion
Amondys51 offers a scientifically backed option for DMD patients whose genetics line up with exon51 skippingdelivering measurable functional gains while demanding careful monitoring. By understanding who qualifies, weighing the proven benefits against the sideeffect profile, and navigating the insurance maze, families can make a confident, informed decision. If youre considering Amondys51, start the conversation with your neuromuscular specialist, request a genetics review, and explore patientassistance resources. Together, we can turn complex science into clearer hope for a steadier, stronger future.
Whats your experience with exonskipping therapies? Share your thoughts in the comments, or reach out if you have any questionslets keep the conversation going!
FAQs
What age groups are eligible for Amondys 51?
Amondys 51 is approved for patients aged 2 years and older who are still ambulatory.
How is Amondys 51 administered?
The medication is given as a weekly intravenous infusion at a qualified infusion center.
Which genetic mutations qualify a patient for Amondys 51?
Eligibility requires a confirmed exon‑51‑amenable DMD mutation, such as deletions, duplications, or point mutations involving exon 51 or neighboring exons.
What are the most common side effects?
Typical side effects include mild infusion‑related reactions like headache, nausea, and temporary increases in serum creatinine.
How can families manage the high cost of Amondys 51?
Manufacturer copay‑assistance programs, nonprofit grants, and prior‑authorization with insurers help offset the roughly $300,000 annual price.