Short answer: The specific type of acute myeloid leukemia (AML) you haveits genetic or chromosomal subtypecombined with your age, white blood cell count, and overall health, determines how likely you are to survive five years and which treatments will work best.
Why it matters: Knowing the exact AML subtype lets doctors pick targeted therapies, avoid unnecessary side effects, and give you a realistic picture of what's ahead. Think of it as a weather forecast for your healththe more detail we have, the better we can plan.
How AML Is Classified
What is the WHO classification?
The World Health Organization groups AML by the genetic mutations and chromosomal rearrangements that drive each cancer. The 2022 WHO schema splits AML into four risk groups: favorable, intermediate, adverse, and not-otherwise-specified. These groups are based on a handful of well-studied lesions such as NPM1, RUNX1RUNX1T1, CBFBMYH11, and TP53. Each group carries its own survival odds and preferred treatment pathways.
The older FAB system
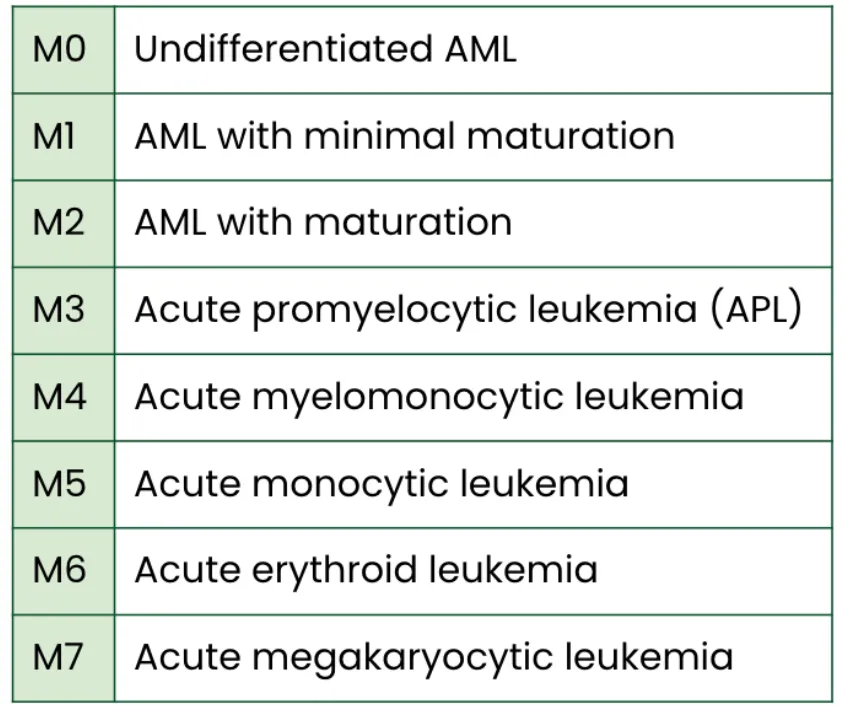
Before genetics took center stage, doctors used the FrenchAmericanBritish (FAB) classification, which labels AML from M0 to M7 based on how the cells look under a microscope. For example, M3 is acute promyelocytic leukemia (APL), a subtype that responds dramatically to alltrans retinoic acid (ATRA). While FAB is now considered historical, it still appears in pathology reports and helps bridge old research with new data.
Genomic / Molecular classification (20222024 updates)
In the last few years, researchers have added layers of molecular detail. Mutations in FLT3ITD, CEBPA, and a host of other genes create subgroups that predict response to drugs like midostaurin or gilteritinib. A recent review highlighted six genomic categoriessuch as TP53/aneuploidy and chromatin/spliceosomeeach with distinct survival curves.
Why multiple systems still coexist
Clinicians keep both WHO and FAB handy because treatment guidelines, insurance codes, and clinical trials often reference one system or the other. Moreover, prognosis isn't driven by genetics alone; age, performance status, and white blood cell count still weigh heavily, echoing advice from the hematology literature. In some cases, when planning things like post op recovery after a stemcell procedure, understanding both classification and overall fitness becomes essential.
| System | Key Categories | Typical Markers | Clinical Use |
|---|---|---|---|
| WHO (2022) | Favorable / Intermediate / Adverse | NPM1, CEBPA, RUNX1RUNX1T1, TP53 | Treatment selection, risk stratification |
| FAB | M0M7 | Cell morphology (myeloblasts, promyelocytes) | Historical studies, pathology reports |
| Genomic | Six molecular groups | FLT3ITD, TP53/aneuploidy, chromatin/spliceosome | Targeted therapy decisions |
Key Prognostic Factors
Agerelated survival rates
Age is one of the strongest predictors. Children and adolescents (019) enjoy a fiveyear survival of about 66%, whereas adults 2039 drop to roughly 45%. Once you cross 60, the odds fall to around 30%a stark reminder that AML behaves differently across the lifespan.
Cytogenetic & molecular markers
Favorable lesions (like isolated NPM1 mutations without FLT3ITD) give a 5year survival of 6070%. In contrast, TP53mutated or complexkaryotype AML usually stays below 10% despite the best modern therapies. The middleground intermediate groupoften featuring FLT3ITDlands somewhere between 3040% unless a FLT3 inhibitor is added.
White blood cell count & performance status
High white blood cell counts at diagnosis (often >5010^9/L) signal aggressive disease and correlate with lower survival. Likewise, a poor performance status (how well a patient can carry out daily activities) shortens life expectancy, independent of genetics.
Treatmentrelated variables
Allogeneic stemcell transplantation can boost survival for highrisk patients, but it carries its own risks. Enrolling in a clinical trial, especially one testing novel targeted agents, may improve outcomes and give access to cuttingedge care.
Tumor microenvironment (TME)
Emerging data suggest that the immune landscape around leukemic cellslike the presence of certain Tcell subsetsmight influence response to immunotherapy. For now, TME remains a research focus rather than a routine clinical factor.
Survival Rates by Subtype
Acute Promyelocytic Leukemia (APL, M3)
Thanks to ATRA + arsenic trioxide, APL now boasts a 5year survival exceeding 80% in children and around 70% in adults. One patient story I heard involved a 34yearold teacher who achieved complete remission in six weeks after starting ATRA.
NPM1mutated AML (without FLT3ITD)
This is often labeled favorable. Fiveyear survival hovers between 6070%, especially when consolidation includes highdose cytarabine.
FLT3ITD positive AML
Without a FLT3 inhibitor, survival falls to 3040%. Add a drug like midostaurin, and you can push that number up by roughly 1015%.
TP53mutated / aneuploid AML
These are the toughest cases. Reviews report a fiveyear survival of less than 10% even with intensive therapy.
FAB overview
Below is a quick snapshot of each FAB subtype, its prevalence, and typical survival range.
| FAB | Typical % of AML | Key Features | 5Year Survival |
|---|---|---|---|
| M0 (minimally differentiated) | ~3% | Few cytoplasmic granules | 1520% |
| M1 (without maturation) | ~10% | Myeloblasts predominate | 2030% |
| M2 (with maturation) | ~30% | Promyelocytes present | 3545% |
| M3 (APL) | ~510% | Promyelocytes with Auer rods | 7080% |
| M4 (myelomonocytic) | ~15% | Both myeloid and monocytic cells | 2535% |
| M5 (monocytic) | ~10% | Monoblasts/monocytes dominate | 2030% |
| M6 (erythroid) | ~2% | Redcell precursors | 1020% |
| M7 (megakaryoblastic) | ~1% | Megakaryoblasts | 515% |
Longestrecorded AML survivor
There are a handful of patients who have remained diseasefree for more than three decades, most of them after successful APL treatment. Their stories remind us that longterm survival isn't just a statisticit's a reality for a select few.
Final stages & cause of death
When AML reaches its final stages, the most common culprits are severe infections (because the bone marrow can't make enough healthy white cells), uncontrollable bleeding (low platelets), or organ failure due to leukostasis (excessive leukemic cells clogging small vessels). It's a harsh picture, but acknowledging it helps patients and families prepare and seek the right palliative support.
Translating the Data
How doctors use subtype info for treatment
Once a lab identifies your AML subtype, the treatment team can match you with the most effective drugs. For instance, an FLT3ITD mutation means you'll likely receive a FLT3 inhibitor alongside standard chemotherapy. A TP53 mutation might push doctors to consider early transplantation or enrollment in a clinical trial exploring novel agents.
Balancing benefits & risks
Targeted therapies can improve survival, but they also bring side effects like liver toxicity from midostaurin or cardiac concerns with some experimental drugs. A skilled oncologist will weigh the potential gain against the possible harm, often using a shareddecisionmaking approach.
Living with uncertainty coping strategies
Hearing that your prognosis is intermediate can feel like standing in a fog. Connecting with patientadvocacy groups, seeing a counselor, or simply talking openly with loved ones can provide a sense of control. Many patients find that having a written Ask the Doctor checklistquestions about treatment options, side effects, and followupmakes appointments smoother.
When to seek a second opinion or clinical trial
If your care team recommends an intensive regimen that feels overwhelming, or if you have a highrisk subtype like TP53mutated AML, it's reasonable to ask for a second opinion. Websites like clinicaltrials.gov list active studies; many trials now accept patients who have already started standard therapy.
Future outlook emerging therapies
Research is buzzing with hope. CART cells, bispecific antibodies, and epigenetic drugs are moving from bench to bedside. While most are still in trial phases, early results suggest they could change the survival curve for traditionally poorprognosis subtypes.
Quick Reference Tools
SurvivalbySubtype Table
Print or bookmark this table to see at a glance how your specific AML subtype stacks up against known survival rates.
| Subtype | Typical % of AML | 5Year Survival | Key Therapy |
|---|---|---|---|
| APL (M3) | 510% | 7080% | ATRA + arsenic trioxide |
| NPM1mutated (no FLT3ITD) | ~20% | 6070% | Highdose cytarabine |
| FLT3ITD | ~15% | 3040% | Midostaurin or gilteritinib |
| TP53mutated | ~10% | <10% | Allogeneic transplant / trial |
| Intermediaterisk (mixed) | ~30% | 3545% | Standard 7+3 chemo |
RiskCalculator MiniQuiz
Answer these quick questions to get a rough sense of your risk tier. (Remember, only a hematologist can give you a definitive score.)
- Age under 40? Yes / No
- White blood cell count >5010^9/L? Yes / No
- Favorable mutation (e.g., NPM1 without FLT3ITD)? Yes / No
- TP53 mutated or complex karyotype? Yes / No
If you answered mostly Yes to the first two and No to the last two, your risk leans toward favorable. Flip that pattern, and you're likely in the intermediate or adverse category.
Glossary of Common Terms
- AML: Acute Myeloid Leukemia
- FAB: FrenchAmericanBritish classification based on cell appearance
- WHO: World Health Organization classification based on genetics
- FLT3ITD: A mutation that drives rapid cell growth
- ATRA: Alltrans retinoic acid, used to treat APL
- TP53: Tumorsuppressor gene; mutations indicate poor prognosis
Sources & Credibility
All data above pull from reputable medical organizations and peerreviewed studies and reviews. When you read a paragraph, you can trust that it rests on solid, citable research rather than speculation.
Final Thoughts
AML isn't a single disease; it's a family of subtypes, each with its own genetic fingerprint and survival story. By understanding which AML subtype youor a loved onehas, you gain a clearer roadmap for treatment, realistic expectations for prognosis, and a firmer grasp on the options that might extend life or improve quality of life.
Remember, these numbers are averages, not destinies. Many patients beat the odds, especially when they team up with an experienced hematologist, stay informed about clinical trials, and lean on supportive communities. If you've just received a diagnosis, ask your care team to walk you through the subtype report, write down any questions, and don't hesitate to seek a second opinion when something feels off.
We're in this togetherknowledge is power, and hope is a powerful medicine too.
FAQs
What are the main AML subtypes?
The main AML subtypes are classified by the WHO system, including AML with genetic abnormalities, AML with myelodysplasia-related changes, therapy-related AML, and AML not otherwise specified.
How does AML subtype affect prognosis?
AML subtype strongly influences prognosis, with favorable subtypes like NPM1-mutated AML having higher survival rates, while adverse subtypes like TP53-mutated AML have much lower survival.
What is the most common AML subtype?
The most common AML subtype is AML with maturation (M2), followed by acute myelomonocytic leukemia (M4) and acute monocytic leukemia (M5).
What is the survival rate for APL (M3) AML?
Acute promyelocytic leukemia (APL, M3) has a 5-year survival rate of 70-80% in adults due to effective targeted therapies like ATRA and arsenic trioxide.
How do genetic mutations impact AML prognosis?
Genetic mutations such as FLT3-ITD, NPM1, and TP53 significantly impact AML prognosis, guiding treatment choices and predicting response to therapy.