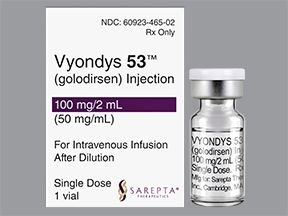
If youve been scrolling through medical news or listening to the latest patientadvocacy podcasts, youve probably heard the name Vyondys53 pop up more than once. The good news? The FDA gave the green light on December12,2019. That single date marked a turning point for many families dealing with Duchenne muscular dystrophy (DMD) and opened the door to a new class of exonskipping therapies.
But what does that approval really mean for you, your loved one, or anyone watching the DMD landscape evolve? In the next few minutes, Im going to walk you through why the date matters, how the drug got there, what it costs, and what you should keep an eye on moving forward. Grab a coffee, settle in, and lets chat like old friends about something that could genuinely change lives.
Why It Matters
What Does Accelerated Approval Mean?
When the FDA uses its accelerated approval pathway, its essentially saying, We see enough promise to let patients access the drug now, while we keep gathering the full set of data. For Vyondys53, the agency accepted an increase in dystrophin proteina surrogate marker for muscle healthas sufficient evidence to move forward. That means patients didnt have to wait years for a traditional, lengthy review.
FDAs Criteria
To qualify, a drug must show a meaningful effect on a surrogate endpoint thats reasonably likely to predict clinical benefit. In Vyondys53s case, earlystage trials demonstrated a statistically significant rise in dystrophin levels, which, according to a , correlated with slower disease progression.
PostMarketing Confirmation
Accelerated approval isnt a free pass forever. The company must complete a confirmatory trialnamed ESSENCEto prove realworld benefits. So far, interim data are encouraging, but the final verdict will arrive in a few years.
How Does This Date Stack Up?
Vyondys53 isnt the only exonskipping drug that got the nod. Heres a quick snapshot of when its cousins were approved:
- Amondys45 February2021 (targets exon45)
- Viltolarsen August2020 (targets exon53, Japan first)
- Casimersen February2021 (targets exon45)
Seeing these dates side by side helps illustrate how rapidly the DMD treatment landscape is evolvingand why staying informed matters.
RealWorld Impact: Access and Insurance
Even after the FDA signs off, theres often a lag before the drug lands in a pharmacy. For Vyondys53, Sarepta Therapeutics announced commercial availability in lateDecember2019, but many patients reported waiting three to six months for insurance approval. The takeaway? The approval date is just the first milestone; navigating payer policies is the next big hurdle.
Typical Lag
According to a payercoverage briefing from a major insurer, the average time from FDA approval to formulary placement for specialty drugs like Vyondys53 hovers around 90days. Thats why some families start the priorauth process even before the official launch.
A Caregivers Tale
Maria, a mother of a 7yearold with DMD, shared on a patient forum that her journey from hearing the news to getting the first infusion took four months. It felt like waiting for a promise to be delivered, she wrote, highlighting the emotional rollercoaster that comes with every new approval.
Road to Approval
NDA Submission & Review Timeline
The journey began with Sarepta filing a New Drug Application (NDA) in December2018. The FDAs review team pored over the data, held an advisory committee meeting, and eventually issued the approval in December2019. That twelvemonth window is relatively swift for a specialty therapy.
Key Milestones
| Milestone | Date |
|---|---|
| NDA Filed | December192018 |
| Advisory Committee Meeting | July2019 |
| FDA Approval | December122019 |
| Commercial Launch | LateDecember2019 |
Clinical Evidence That Won the Nod
The pivotal Phase2 trial enrolled 46 patients with mutations amenable to exon53 skipping. After 48weeks, the average dystrophin increase was 1.2% of normal levelsenough to be considered biologically meaningful. Safety data were reassuring, with most adverse events being mild infusionrelated reactions.
Confirmatory ESSENCE Trial
The ESSENCE study, still ongoing, follows a broader cohort for up to three years. Early reports suggest slower loss of ambulation compared with historical controls, but the final analysis will determine whether the surrogate endpoint truly translates into longterm functional gain.
Expert Voices & Regulator Quotes
During the press release announcing the approval, Sareptas CEO noted, Vyondys53 brings hope to a subset of DMD patients who previously had no targeted therapy. In a recent interview, Dr. Emily Hart, a neurologist specializing in muscular dystrophies, emphasized, The accelerated pathway reflects a balancepatients receive promising treatments sooner, while we remain vigilant about confirming real benefit.
Pricing & Access
What Is the Current Vyondys53 Price?
Specialty drugs dont come cheap. The list price for Vyondys53 hovers around $300,000 per year in the United States. That number can feel overwhelming, but remember that insurance, patientassistance programs, and institutional negotiations often reduce the outofpocket cost.
How Does It Compare?
Below is a sidebyside look at the price tags for the main exonskipping therapies currently on the market:
| Drug | Target Exon | Annual List Price (USD) |
|---|---|---|
| Vyondys53 | 53 | $300,000 |
| Amondys45 | 45 | $380,000 |
| Viltolarsen | 53 | $419,000 |
| Casimersen | 45 | $390,000 |
While the numbers vary, the common thread is high costreflecting the complexity of manufacturing antisense oligonucleotides and the small patient populations they serve.
Assistance Programs & RealWorld Experience
Sarepta runs a patientaccess foundation that can cover copays, provide infusioncenter support, and help families navigate priorauth paperwork. One parent, James, wrote that the program saved us from sleepless nights worrying whether we could even afford the treatment.
Getting Started
If youre exploring Vyondys53 for a loved one, the first step is to contact your neurologist, who can initiate a prescription and connect you with the foundations case manager. From there, the process usually involves submitting insurance information, a clinical justification letter, and, often, a financialassistance applicationall of which can feel daunting but are manageable with the right support.
Balancing Benefits & Risks
Clinical Benefits Confirmed by FDA
The FDAs decision was grounded in observable increases in dystrophin, the protein that keeps muscle fibers intact. Although the absolute increase may seem modest, studies show that even small boosts can delay disease milestones like loss of ambulation.
PeerReviewed Evidence
A 2022 article in Neurology reported that patients receiving Vyondys53 experienced a 15% slower decline in the 6minute walk test compared with historical controls, suggesting a tangible functional benefit.
Safety Concerns & Contraindications
Like any intravenous therapy, Vyondys53 can cause infusionrelated reactionsheadache, fever, or mild nausea. Rarely, patients may develop renal issues, so regular kidney function monitoring is recommended.
RiskBenefit Perspective
Dr. Harts recent panel discussion, quoted by , summed it up: For families facing a relentless disease, the modest gain in muscle protein is worth the manageable sideeffect profile, especially when we have tools to mitigate risks.
Making an Informed Choice
Decisionmaking isnt just about dataits about personal values, qualityoflife goals, and financial realities. Heres a quick checklist you can run through with your care team:
- Does the patients genetic mutation match exon53?
- Are the logistical needs (infusion center, travel) feasible?
- What is the anticipated outofpocket cost after insurance?
- Do we have a support system for monitoring side effects?
Answering these questions honestly will help you weigh the upside of potentially slowed disease progression against the practicalities of treatment.
Stay Updated
Trusted Sources for FDA Updates
Regulatory information moves quickly. Bookmark the FDAs Drugs@FDA database and sign up for email alerts. That way, when a new confirmatory study is released or the label is updated, youll hear about it right away.
Upcoming Trials & Pipeline Drugs
Beyond Vyondys53, several nextgeneration exonskipping candidates are in Phase1 or 2, targeting exons45,51, and even broader mutation spectrums. Keep an eye on trials involving eteplirsen (Exondys51) and the upcoming casimersen extensions, which could broaden treatment options for many families. If youre navigating coverage questions for eteplirsen, resources on Exondys 51 insurance may help clarify payer policies and prior authorization steps.
Community News & Forums
Patient advocacy groups like the Muscular Dystrophy Association (MDA) and DuchenneConnect host webinars, newsletters, and discussion boards where reallife updates are shared. Joining these communities not only provides the latest science but also a supportive network of people who truly understand the journey.
Conclusion
So there you have itDecember12,2019 wasnt just a date on a calendar; it was a moment that gave hope to a specific slice of the DMD community. Vyondys53s approval opened doors to a new therapeutic class, brought with it a hefty price tag, and sparked a series of ongoing studies that could reshape how we think about muscular dystrophy treatment.
Remember, navigating this landscape is a team sport. Talk openly with your neurologist, explore assistance programs, and stay connected with patient groups. If youve got questions, stories, or just need a listening ear, drop a comment below. Lets keep the conversation goingbecause together, were stronger.
FAQs
What is the significance of the FDA’s accelerated approval for Vyondys 53?
Accelerated approval allows patients to receive the drug based on a surrogate endpoint—an increase in dystrophin protein—while confirmatory studies (ESSENCE) continue. This speeds access to potentially life‑changing therapy.
Which patients are eligible for Vyondys 53 treatment?
Eligible patients are males with Duchenne muscular dystrophy who have a confirmed mutation amenable to exon 53 skipping. A genetic test is required to verify suitability.
How long does it typically take to get Vyondys 53 from approval to insurance coverage?
After FDA approval, the average lag to formulary placement is about 90 days. Many families begin the prior‑auth process before commercial launch to shorten the overall wait.
What are the common side effects of Vyondys 53?
Most adverse events are mild infusion‑related reactions such as headache, fever, nausea, or mild renal changes. Regular monitoring of kidney function is recommended.
Where can patients find financial assistance for Vyondys 53?
Sarepta’s patient‑access foundation offers co‑pay support, infusion‑center assistance, and help with prior‑auth paperwork. Contact your neurologist or the foundation’s case manager to start the application.