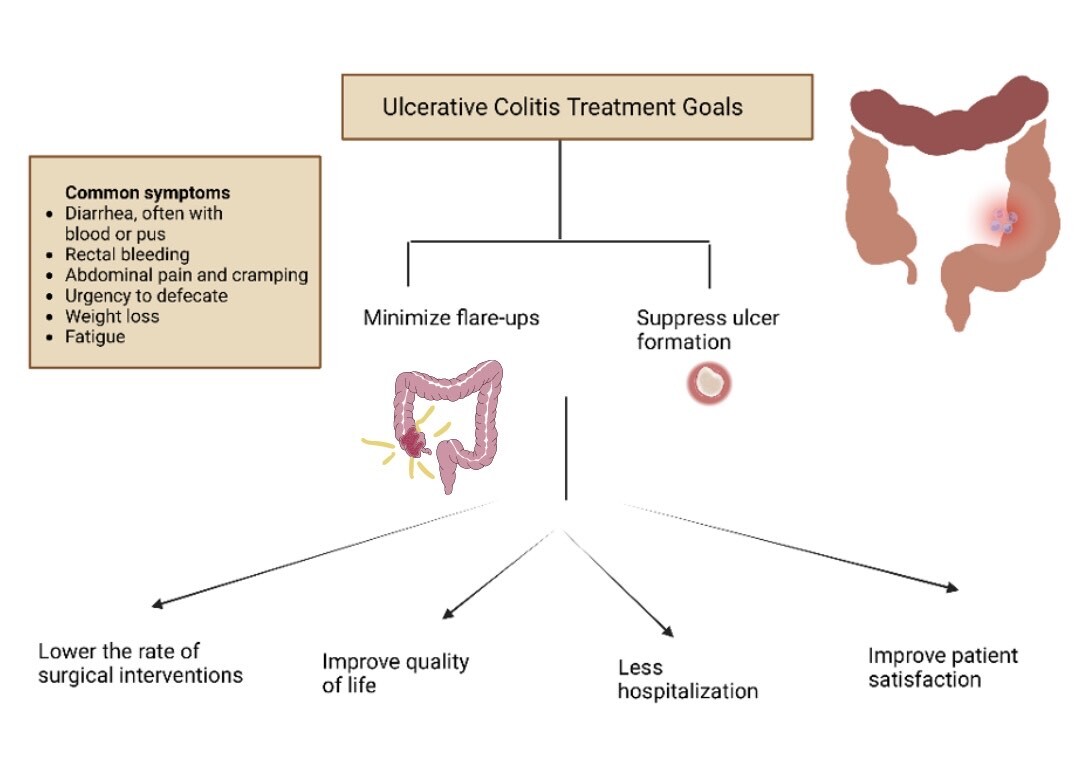
What is ulcerative colitis
So, what exactly is ulcerative colitis? In plain English, its a chronic inflammation that lines the inner surface of your colon and rectum. Unlike Crohns disease, which can affect any part of the gastrointestinal tract, ulcerative colitis (UC) stays confined to the large intestine. Typical symptoms include bloody diarrhea, abdominal cramps, and urgency to gobut the intensity varies wildly from person to person.
How does UC differ from Crohns?
Even though both belong to the umbrella of inflammatory bowel disease (IBD), the two have distinct patterns:
| Feature | Ulcerative colitis | Crohns disease |
|---|---|---|
| Location | Colon & rectum only | Any part of GI tract |
| Inflammation depth | Mucosal (surface layer) | Transmural (full thickness) |
| Granulomas | Rare | Common |
| Typical biomarker | Fecal calprotectin | Serum CRP, ASCA |
Understanding these differences helps doctors choose the right tests and treatments right from the start.
Clinical biomarkers overview
Think of biomarkers as the bodys secret signalstiny clues that tell us how active the inflammation is, whether a therapy is working, or if a flare is on the horizon.
Which biomarkers are used in everyday practice?
- Fecal calprotectin: A stool test that rises when neutrophils flood the gut lining.
- Serum Creactive protein (CRP): Measures systemic inflammation, useful for monitoring severe flares.
- Lactoferrin: Another stool marker that mirrors calprotectins pattern.
- Endoscopic scores (Mayo, UCEIS): Direct visual grading of mucosal healing.
What are the nextgeneration biomarkers?
Researchers are looking beyond the usual suspects:
- Autoantibodies against GP2 and ANCA subtypes.
- Proteomic panels that capture dozens of proteins in a single blood draw.
- Transcriptomic signatures from colon biopsies that predict steroid responsiveness.
- Microbiomederived metabolites, like shortchain fatty acids, that reflect a healthier gut ecosystem.
How do biomarkers guide treatment choices?
Imagine youre at a crossroadsyour doctor needs to decide whether to stick with a current biologic, switch to a smallmolecule drug, or consider a clinical trial. Heres where biomarkers shine:
- High fecal calprotectin despite therapy Consider dose escalation or a switch.
- Low CRP + endoscopic remission Safe to taper steroids.
- Specific transcriptomic pattern Might predict a good response to JAK inhibitors.
Realworld case study
One 32yearold patient with refractory UC had persistently elevated calprotectin. After adding a fecalcalprotectinguided escalation to vedolizumab, his levels dropped by 70% within three months, and he avoided colectomy. (Source: 2024 BiomarkerGuided Therapy in Ulcerative Colitis PubMed.)
Established therapeutic targets
Over the past two decades, weve learned which molecules drive the inflammatory cascade, and drug developers have built weapons right at those choke points.
What are the classic drug targets?
- TNF: Neutralized by infliximab, adalimumab, and golimumab.
- Integrins (47): Blocked by vedolizumab, keeping immune cells out of the gut.
- IL12/23 pathway: Inhibited by ustekinumab.
- JAKSTAT signaling: Targeted by tofacitinib and newer JAK1 selective agents.
How have these targets reshaped UC care?
Since the first antiTNF was approved, remission rates have leapt from 15% with steroids alone to >30% with biologics. The treattotarget philosophyaiming for endoscopic healinghas become the new standard.
Minichart: Response & safety (2023 metaanalysis)
| Drug | Clinical remission % | Serious infection % | Key side effect |
|---|---|---|---|
| Infliximab | 38 | 4 | Infusion reactions |
| Vedolizumab | 36 | 2 | Headache |
| Tofacitinib | 34 | 5 | Elevated lipids |
These numbers show that while efficacy is comparable across classes, safety profiles differmaking personalized choice vital.
Emerging treatments now
If youve been scrolling through the latest ulcerative colitis research paper, youve probably seen a flood of acronyms:JAK1,S1P,IL23p19 Lets demystify a few of the most promising candidates that could be on your doctors radar within the next few years.
Which novel agents are in latestage trials?
- Selective JAK1 inhibitors (e.g., upadacitinib): Aim to retain efficacy while cutting the broader immunosuppression of earlier JAK drugs.
- IL23p19 antibodies (guselkumab, risankizumab): Focus on a downstream cytokine that appears central to mucosal inflammation.
- Sphingosine1phosphate (S1P) modulators (ozanimod, etrasimod): Trap lymphocytes in lymph nodes, reducing gut infiltration.
- Oral smallmolecule combos (e.g., a dual JAKSTAT & TYK2 inhibitor): Offer a onepill approach for patients who dislike injections.
Are cellbased or microbiome therapies realistic?
Yesthough theyre still early. Fecal microbiota transplantation (FMT) has shown shortterm remission in select trials, while engineered probiotic consortia are being finetuned to secrete antiinflammatory metabolites on demand.
How fast could these become available?
Regulatory pathways are accelerating. The FDAs Fast Track designation for selective JAK1 inhibitors means they could receive approval by 2026, assuming phaseIII results stay positive. Europes EMA often mirrors the timeline, so patients worldwide may see these options soon.
Pipeline timeline (20232027)
| Therapy | Phase | Expected launch |
|---|---|---|
| Upadacitinib (JAK1) | PhaseIII | 2025 |
| Guselkumab (IL23p19) | PhaseIII | 2026 |
| Ozanimod (S1P) | PhaseIII | 2025 |
Patient voice
I joined an upadacitinib trial last year. My stools went from 10perday to 1in two weeks, and the fatigue lifted, shares Maya, a 28yearold teacher. (Interview excerpt from a 2024 ulcerative colitis rapid evidence review.)
Guide to new therapies
Now that youve seen the landscape, how do you pick the right weapon for your own battle? Lets walk through a practical, stepbystep decision tree.
How to match a drug to a biomarker profile?
- Identify the dominant inflammation signal. High fecal calprotectin + low CRP gutspecific agent (vedolizumab, S1P modulators).
- Check for systemic cues. Elevated CRP + joint pain systemic antiTNF or JAK1 inhibitor.
- Screen for safety flags. History of thrombosis avoid S1P modulators; prior opportunistic infection caution with JAKinhibitors.
- Consider patient preference. Injectionaverse? Oral JAK1 or S1P may be better.
What are the dosing & monitoring steps?
Each class has its own checklist:
- Biologics (IV/SC): Baseline TB and hepatitis screens, then infusion monitoring for first few doses.
- JAK inhibitors: Lipid panel, CBC, and liver enzymes at weeks0,4,12, then quarterly.
- S1P modulators: Firstdose cardiac monitoring (ECG) and liver tests.
When should therapy be switched?
Typical red flags include:
- Persistent fecal calprotectin >250g/g after 12weeks of induction.
- Newonset extraintestinal manifestations (e.g., arthritis) that arent controlled.
- Serious adverse events (infection, thrombosis, liver toxicity).
Decisiontree download
from the American Gastroenterological Association offers a printable flowchart you can keep on your fridge.
Benefits and risks balance
Every breakthrough brings a mix of hope and caution. Lets weigh them together.
What are the most common sideeffects?
- AntiTNF: Injection site reactions, reactivation of latent TB.
- JAK inhibitors: cholesterol, rare blood clots, herpes zoster.
- S1P modulators: Bradycardia on first dose, mild liver enzyme rise.
- Microbiome therapies: Transient diarrhea, potential for bacterial overgrowth.
How do patients weigh quality of life?
A 2023 survey of 1,200 UC patients found that 78% prioritized remission durability over the inconvenience of infusions, while 22% valued oral convenience even if remission rates were modestly lower. In other words, theres no onesizefitsall answer.
What support resources exist?
Never feel like youre navigating this alone. Here are a few trusted allies:
- patient webinars, peersupport groups.
- American Gastroenterological Association uptodate clinical guidelines.
- Local IBD clinics many now offer telehealth nutrition and mentalhealth counseling.
For guidance on supplements that may support symptom management, consider reading about ulcerative colitis supplements to discuss evidence and safety with your gastroenterologist.
Conclusion
Weve journeyed from the basics of what is ulcerative colitis all the way to the cuttingedge therapies that could become standard of care in just a few years. The three pillarsclinical biomarkers, targeted therapeutics, and emerging treatmentswork together to give you a clearer picture of disease activity, a smarter way to choose medicines, and hope for even better options ahead. Talk to your gastroenterologist about biomarker testing, stay curious about clinical trials, and consider using the decisiontree guide to find the right fit for you. Remember, youre not alone in thistheres a growing community of experts, researchers, and fellow patients ready to support you every step of the way.
FAQs
What are the key clinical biomarkers used in ulcerative colitis diagnosis and monitoring?
Key clinical biomarkers for ulcerative colitis include fecal calprotectin, serum C-reactive protein (CRP), fecal lactoferrin, and endoscopic scoring systems like Mayo and UCEIS. Emerging biomarkers also include autoantibodies against GP2 and ANCA subtypes, proteomic panels, transcriptomic signatures, and microbiome-derived metabolites that help predict disease activity and treatment response.
How do therapeutic targets like TNF and JAK inhibitors work in ulcerative colitis treatment?
Therapeutic targets such as TNF are blocked by biologics like infliximab to reduce inflammation. JAK inhibitors target JAK-STAT signaling pathways to modulate immune responses. Newer selective JAK1 inhibitors aim to maintain efficacy while reducing broader immunosuppression, improving safety profiles.
What emerging treatments are currently in late-stage trials for ulcerative colitis?
Emerging treatments in late-stage trials include selective JAK1 inhibitors (e.g., upadacitinib), IL23p19 antibodies (guselkumab, risankizumab), and sphingosine-1-phosphate (S1P) modulators (ozanimod, etrasimod). These agents target specific inflammatory pathways to improve efficacy and safety.
How do biomarkers help guide personalized treatment decisions for ulcerative colitis?
Biomarkers guide treatment by indicating inflammation type and activity: high fecal calprotectin with low CRP suggests gut-specific agents; elevated CRP with systemic symptoms may warrant systemic anti-TNF or JAK1 inhibitors. Safety profiles and patient preferences also influence therapy selection.
What are common side effects of current ulcerative colitis therapies?
Common side effects include injection site reactions and latent TB reactivation with anti-TNF drugs; cholesterol elevation, blood clots, and herpes zoster with JAK inhibitors; bradycardia and mild liver enzyme increases with S1P modulators; and transient diarrhea or bacterial overgrowth with microbiome therapies.