Therapyrelated acute myeloid leukemia (tAML) isnt just a fancy term you see in a textbook its a real, often frightening reality that can pop up after chemo, radiation, or other intense treatments. The good news is that a handful of genetic mutations, known as therapyrelated AML mutations, drive this disease, and understanding them can spell the difference between uncertainty and a clear, actionable plan.
Knowing which mutations are most common, how they shape prognosis, and what targeted therapies exist helps you (or a loved one) make informed decisions, talk confidently with doctors, and feel a little less alone in a tough journey.
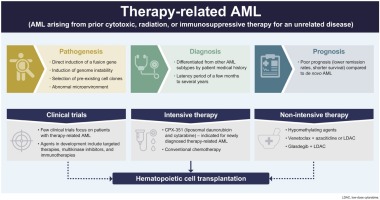
What Is TherapyRelated AML?
Definition and latency period
tAML is a form of acute myeloid leukemia that arises as a late complication of previous cancer therapy. It typically shows up 210years after exposure to alkylating agents, topoisomeraseII inhibitors, or highdose radiation. The disease is secondary because the DNA damage from those treatments creates new mutations that eventually drive malignant growth.
Who gets it?
Although tAML accounts for roughly 510% of all AML cases, its incidence climbs in survivors of breast cancer, lymphoma, and multiple myeloma. Older adults (over 60) and those who received highdose chemo are at the highest risk.
Why does it matter?
Therapyrelated AML often carries a poorer outlook than denovo AML because the mutations tend to be more aggressive. But acknowledging those risks early opens the door to precision medicine, clinical trials, and, in some cases, curative transplant.
Key Mutations in tAML
| Mutation | Frequency in tAML* | Typical Clinical Impact | Related Keywords |
|---|---|---|---|
| TP53 | 3040% | Complex karyotype, resistance to standard chemo, poor prognosis | therapy-related AML prognosis |
| NPM1 | 45% | Often favorable when isolated, but less so in tAML | therapy-related AML cytogenetics |
| FLT3ITD | 23% | Higher relapse risk; targetable with FLT3 inhibitors | therapy-related AML mutations |
| DNMT3A | 28% | Epigenetic dysregulation; modest impact on survival | therapy-related AML mutations |
| ASXL1 / RUNX1 | 1015% each | Associated with adverse cytogenetics | therapy-related AML prognosis |
*Data compiled from a 2024 molecular landscape study of secondary and therapyrelated AML (PMCID8610791).
Why TP53 is the bad actor
TP53 acts like the cells security guard. When its knocked out, damaged DNA roams free, leading to a chaotic, often therapyresistant leukemia. Studies show TP53mutated tAML patients have a median overall survival of just 912months, even with aggressive treatment.
NPM1mutated tAML vs. denovo NPM1 AML
In denovo AML, an isolated NPM1 mutation often predicts a better response to standard chemo. In tAML, however, NPM1 is frequently accompanied by other highrisk lesions (like TP53 loss), which blunts its usual advantage.
Emerging mutations and their relevance
Newer sequencing panels are flagging IDH1/2, NRAS, and KRAS as occasional players in tAML. Those mutations matter because FDAapproved IDH inhibitors (enasidenib, ivosidenib) can be added to a tailored regimen, offering a glimmer of hope for patients who otherwise have limited options.
Cytogenetics and Risk
Common cytogenetic abnormalities
Complex karyotype (3 chromosomal abnormalities) and monosomal karyotype are hallmarks of therapyrelated AML. These patterns often coexist with TP53 loss, cementing the diseases aggressive nature.
ELN 2022 risk stratification for tAML
The European LeukemiaNet (ELN) integrates both mutations and cytogenetics to sort patients into Favorable, Intermediate, or Adverse risk categories. For tAML, the Adverse group swells because many patients carry TP53 mutations and complex karyotypes.
| Risk Group | Typical Mutation/Cytogenetics | Example Scenario |
|---|---|---|
| Favorable | NPM1 isolated, normal karyotype | 30yearold lymphoma survivor, good performance status |
| Intermediate | FLT3ITD with normal karyotype | 55yearold breastcancer survivor, moderate comorbidities |
| Adverse | TP53 loss + complex karyotype | 68yearold multiplemyeloma survivor, frail |
Prognosis and Life Expectancy
Overall survival statistics
Across large registries, median overall survival for tAML hovers around 12months, but it stretches to 23years for patients with favorable genetics (e.g., isolated NPM1) and drops below 6months for those harboring TP53 mutations.
Factors that worsen outlook
Age over 60, poor performance status (ECOG2), prior organ toxicity from cancer therapy, and adverse cytogenetics all pull the survival curve down.
tAML life expectancy by mutation type
For a quick snapshot:
- TP53mutated: 612months
- FLT3ITD: 1014months (improved with FLT3 inhibitors)
- NPM1 isolated: 2030months (if no TP53 comutation)
- IDHmutated: 1218months (potentially extended with IDH inhibitors)
Case vignette: a realworld example
Imagine Jane, a 58yearold breastcancer survivor. Two years after finishing adjuvant chemo, she develops fatigue and bruising. Bonemarrow biopsy reveals TP53mutated tAML with a complex karyotype. She was offered a clinical trial combining a hypomethylating agent with venetoclax, but her disease progressed quickly, underscoring the grim prognosis often tied to TP53. Stories like Janes remind us why early genetic testing is crucial.
How to Diagnose tAML
When to suspect tAML
Newonset anemia, unexplained bruising, frequent infections, or a rising blast count on routine blood work after cancer therapy should raise red flags. A quick CBC with differential can be the first clue.
Bonemarrow workup steps
1. Morphology look for myeloblasts >20%.
2. Flow cytometry identify AMLdefining antigens (CD13, CD33, CD117).
3. Nextgeneration sequencing (NGS) panel captures TP53, NPM1, FLT3, DNMT3A, and other actionable lesions.
4. Cytogenetic karyotyping reveals complex or monosomal patterns.
Therapyrelated AML ICD10 coding
The official code is C92.0 with an additional secondary to chemotherapy qualifier. Accurate coding matters not just for billing but also for epidemiologic tracking and research.
Treatment Options Overview
Standard induction (7+3) and its limits
The classic regimenseven days of cytarabine plus three days of an anthracyclinestill forms the backbone of AML treatment. However, TP53mutated patients often fail to achieve complete remission, prompting clinicians to look beyond the standard.
Targeted agents by mutation
FLT3 inhibitors (midostaurin, gilteritinib) improve outcomes for FLT3ITD patients, especially when paired with a hypomethylating agent.
IDH inhibitors (enasidenib for IDH2, ivosidenib for IDH1) have shown durable responses in IDHmutated tAML.
Venetoclax plus azacitidine or decitabine offers a chemofree approach that can yield remission in older or unfit patients, even with adverse genetics.
Allogeneic stemcell transplant (alloSCT)
For eligible patients (typically <60years, good organ function), alloSCT remains the only potentially curative option. The decision hinges on disease remission status, donor availability, and individual risk tolerance.
Decisiontree for transplant feasibility
If you achieve a complete remission (CR) after induction, evaluate: age <60? donor match available? comorbidities low? proceed to transplant. If any answer is no, discuss maintenance strategies or clinical trial enrollment.
Special Populations Affected
Therapyrelated AML in children
Although rare, pediatric tAML frequently follows treatment for acute lymphoblastic leukemia (ALL). The mutational spectrum leans more toward KMT2A rearrangements rather than TP53, and outcomes can be slightly better when aggressive pediatric protocols are applied.
Therapyrelated myeloid neoplasm vs. denovo AML
Both entities share overlapping features, but tAML often carries a higher burden of chromosome abnormalities and a distinct clinical history. Recognizing the therapyrelated label guides clinicians toward more intensive monitoring and, sometimes, different clinical trial eligibility.
Impact of prior immunosuppressive therapy
Patients who received longterm immunosuppression (e.g., after organ transplant) may develop tAML with unique epigenetic changes. Their treatment options are complicated by the need to balance graft rejection risk with aggressive leukemia therapy.
Balancing Benefits and Risks
Why mutation profiling matters for shared decisionmaking
Imagine youre sitting across from your oncologist, a spreadsheet of mutations in front of you. Knowing that your leukemia harbors a FLT3ITD mutation instantly opens the door to an FDAapproved FLT3 inhibitoran option you might not have considered otherwise.
Psychosocial impact of a tAML diagnosis
Its normal to feel a swirl of emotionsanger at the therapy that healed you, guilt about burdening family, and fear of the unknown. Connecting with support groups, counseling services, and survivorship programs can alleviate some of that weight.
Resources and support
Organizations like the Leukemia & Lymphoma Society offer peertopeer mentoring, while clinicaltrial registries (e.g., ) help you stay informed about cuttingedge options. For patients also navigating other cancer survivorship issuessuch as concerns about prostate cancer prognosisinformation on prostate cancer outlook can be useful background when discussing overall life expectancy and survivorship planning with your care team.
Conclusion
Therapyrelated AML mutations turn a prior cancer victory into a new, daunting challenge, but they also supply a roadmap. By recognizing the common culpritsTP53, NPM1, FLT3, DNMT3Aand understanding how they shape cytogenetics, prognosis, and treatment, you gain power over uncertainty. Whether youre navigating a diagnosis yourself, supporting a loved one, or simply seeking knowledge, stay proactive: get comprehensive genetic testing, discuss targeted options with your hematologist, and lean on trusted resources.
Remember, youre not alone in this journey. If you have questions or want to share your experience, feel free to reach outknowledge shared is hope multiplied.
FAQs
What are therapy-related AML mutations?
They are genetic mutations that arise after prior cancer therapy, such as chemotherapy or radiation, which cause DNA damage leading to therapy-related acute myeloid leukemia (tAML).
Which mutations are most common in therapy-related AML?
TP53 mutations occur in 30-40% of tAML cases and are linked to poor prognosis. Other common mutations include NPM1, FLT3ITD, DNMT3A, ASXL1, and RUNX1.
How do therapy-related AML mutations affect prognosis?
Mutations like TP53 are associated with aggressive disease, resistance to standard chemotherapy, and shorter survival, whereas isolated NPM1 mutations may have a more favorable outlook.
Can therapy-related AML mutations be targeted with specific treatments?
Yes. FLT3ITD mutations are targetable with FLT3 inhibitors, and IDH1/2 mutations can be treated using FDA-approved IDH inhibitors, improving patient outcomes.
Why is genetic testing important for patients with therapy-related AML?
Genetic testing identifies specific mutations that guide prognosis assessment and personalized treatment decisions, including eligibility for targeted therapies and clinical trials.