Discovering that acute myeloid leukemia (AML) has come back is like hearing a flat tire after a long road tripyou want a clear fix, not a vague promise. The short answer? A blend of salvage chemotherapy, targeted drugs based on your leukemias genetics, and often a stemcell transplant if youre a good candidate. Below well walk through the main protocols, weigh the benefits and risks, and give you practical steps to keep the disease from striking again.
Understanding Relapse Prognosis
What defines a relapsed AML?
In the medical world, a relapse is declared when bonemarrow blasts rise above 5% or when leukemia cells reappear in the blood after a period of remission. Timing matters tooearly relapse (within six months) usually carries a tougher outlook than a later one.
How is relapsed AML prognosis calculated?
Doctors look at several factors: age, specific genetic mutations like FLT3ITD or IDH, how you responded to the first round of therapy, and whether youve already had a stemcell transplant. These pieces combine into a score that predicts AML relapse life expectancy, which on average ranges from 6 to 12 months, but can stretch beyond two years for younger patients with favorable genetics.
Realworld snapshot
Take Mark, a 52yearold who relapsed four months after his initial treatment. Because he carried a FLT3ITD mutation and responded well to the first therapy, his oncologist opted for a FLT3targeted drug as a bridge to a second transplant. Sixteen months later hes still in remissiona reminder that individual details can shift the odds dramatically.
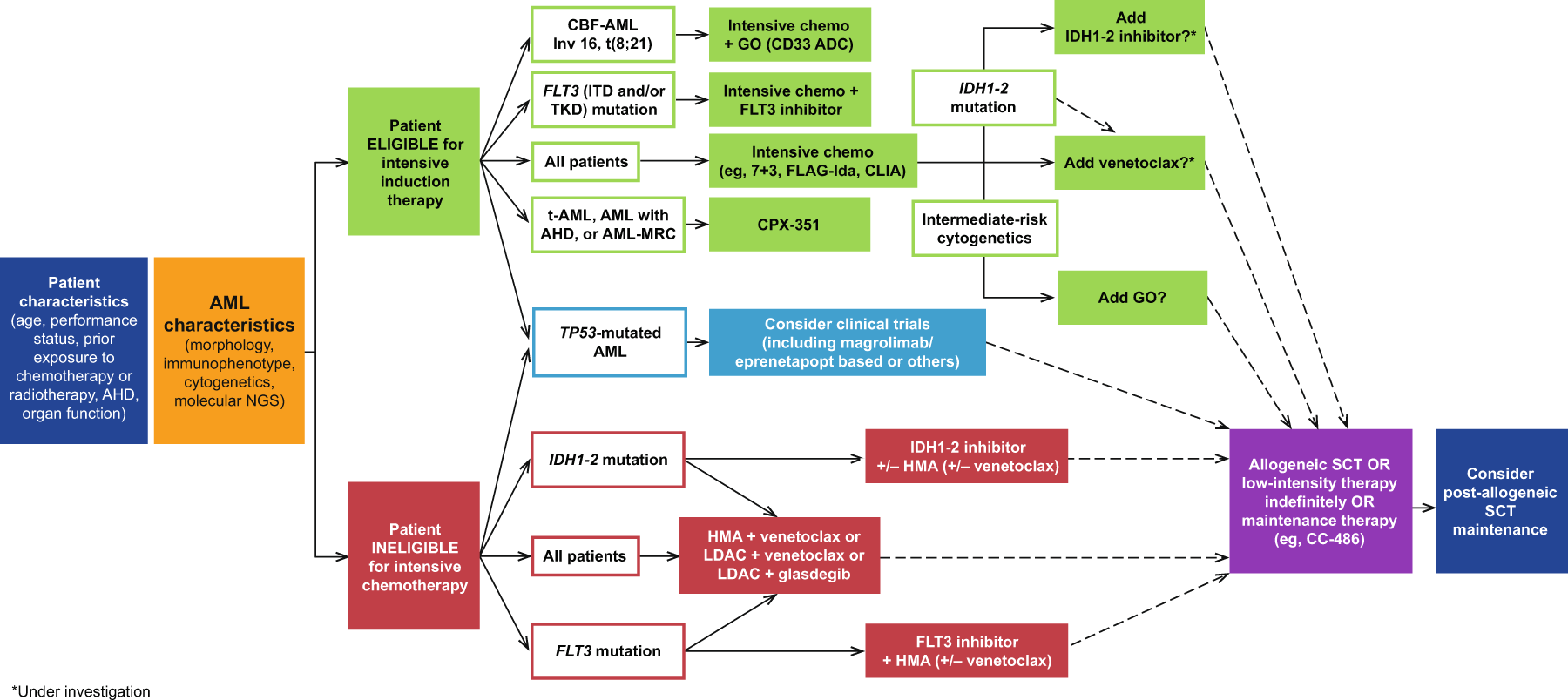
FirstLine Salvage Options
FLAGIDA: intensive and often effective
FLAGIDA combines fludarabine, highdose cytarabine, idarubicin, and GCSF. Its a favorite for patients who can tolerate a hospital stay because it can push remission rates up to 55%. The downside? Its tough on the marrow, so expect a period of low blood counts and infection risk.
MEC: when anthracycline exposure is a concern
MEC (mitoxantrone, etoposide, cytarabine) offers a solid alternative, especially if kidney function is shaky or youve already received a lot of anthracyclines. Response rates hover around 4050%, and its a bit gentler on the heart.
Lowintensity salvage: azacitidinevenetoclax
For older or frailer patients, a combination of the hypomethylating agent azacitidine with the BCL2 inhibitor venetoclax has become a gamechanger. Recent realworld data show overall response rates near 50% with a more manageable sideeffect profile, though prolonged neutropenia can still be an issue.
| Regimen | Typical Setting | CR Rate | Inpatient? | Median OS (months) |
|---|---|---|---|---|
| FLAGIDA | Fit, <30y | 4555% | Yes | 1014 |
| MEC | Renal concerns | 4050% | Yes | 812 |
| Azacitidine+Venetoclax | 60y or unfit | 50% | No (outpatient) | 710 |
Where to learn more
For the most uptodate regimen details, the provide a thorough, evidencebased overview.
Targeted & Novel Agents
FLT3mutated relapse: gilteritinib
If your leukemia carries a FLT3 mutation, gilteritinib is often the first targeted drug of choice. The ADMIRAL trial showed a complete remission (CR) rate of about 21%, and many patients use it as a bridge to transplant.
IDH1/2 mutations: ivosidenib & enasidenib
These oral agents work by forcing the leukemia cells to differentiate rather than proliferate. Pairing them with a hypomethylating agent can boost response rates, but watch for differentiation syndromea feverish, inflammatory reaction that needs prompt steroids.
CD33targeted therapy: gemtuzumab ozogamicin
Gemtuzumab can be given alone or with cytarabine for patients who express CD33. It offers a modest CR boost, though liver toxicity (venoocclusive disease) remains a concern.
Emerging immunotherapies
CART cells and bispecific antibodies are on the horizon for relapsed/refractory AML. Earlyphase trials in 2024 report encouraging remission signals, but these approaches are still experimental and usually limited to academic centers.
Expert insight
According to a recent review in , combining targeted agents with traditional chemotherapy is rapidly becoming the new standard for many genetic subtypes of relapsed AML.
StemCell Transplant Role
When does relapse after transplant happen?
Relapse can be molecular (detectable mutations but no blood count changes) or hematologic (fullblown return of blasts). Molecular relapses are caught earlier with sensitive testing and often have a better chance of being salvaged.
Best bridge to a second transplant
Intensive regimens like FLAGIDA can quickly reduce disease burden, while FLT3inhibitors such as gilteritinib are preferred for FLT3mutated patients because theyre less toxic and still effective. The goal is to achieve a second remission before the conditioning regimen for the transplant.
Risks and rewards of a second transplant
Going through a second allogeneic transplant is a highstakes decision. Nonrelapse mortality can reach 2030%, but for those who make it, longterm survival can exceed 40%especially if a matched sibling donor is available.
Managing Toxicities & Support
Common sideeffects of salvage therapy
Expect bonemarrow suppression, mucositis, and possible liver or kidney strain. Growthfactor support (e.g., filgrastim) and transfusion thresholds help keep you stable while your body recovers.
Infection prophylaxis
During intense chemo, doctors usually prescribe levofloxacin for bacterial coverage, posaconazole for fungal protection, and either acyclovir or valacyclovir to ward off herpesrelated infections.
Psychosocial and financial support
Facing another round of treatment can be emotionally draining. Leverage counseling services, patientadvocacy groups like the Leukemia & Lymphoma Society, and hospital social workers to navigate insurance and medication costs.
Preventing Future Relapse
Maintenance therapy after remission
Oral azacitidine (CC486) and FLT3 inhibitors are now approved for maintenance in certain highrisk patients, extending diseasefree survival by several months. Regular molecular MRD (minimal residual disease) testing helps catch tiny disease reservoirs before they grow.
Lifestyle and followup tips
Stay uptodate on vaccinations (influenza, COVID19, pneumococcal), practice good hand hygiene, and maintain a balanced diet with moderate exercise. Stressmanagement techniqueslike mindfulness or gentle yogacan also contribute to overall resilience.
Clinical trials: why they matter
When standard options feel limited, a trial might offer access to novel agents or combination strategies not yet widely available. Ask your oncologist about active studies; many are recruiting patients with relapsed AML across the country.
Action Plan For You
Step1: Gather your latest bonemarrow pathology report and any genetic sequencing results.
Step2: Schedule a multidisciplinary review with your hematologist, transplant specialist, and a genetics counselor.
Step3: Discuss whether an intensive salvage regimen (like FLAGIDA) or a lowintensity approach with venetoclax is best for you, based on age, organ function, and mutation profile.
Step4: Ask about eligibility for a stemcell transplant or maintenance therapybeing proactive can open doors to lifeextending options. For information on pregnancy-safe options and coordination of AML care during pregnancy consult resources on acute myeloid leukemia pregnancy to help guide discussions when pregnancy is a factor in treatment planning.
Step5: Set up supportivecare measures nowantibiotic prophylaxis, transfusion plans, and access to counseling.
Conclusion
Relapsed AML is undeniably tough, but a clear treatment roadmapstarting with the right salvage chemotherapy, adding geneticsdriven targeted drugs, and considering a timely stemcell transplantoffers real hope for a second remission and longer survival. Understanding both the upside and the risks lets you make informed, confident choices. If you or a loved one is facing this crossroads, take these steps, ask about clinical trials, and lean on the expert team around you. Youre not alone; the medical community, patient groups, and countless survivors are all here to help you navigate the road ahead.