Stick with me, and youll come away with a clear roadmap: a quick way to spot relapse symptoms, a rundown of the most common relapsed AML treatment protocols, realistic expectations for life expectancy, and practical steps to keep AML from sneaking back in again.
Understanding Relapse
What Relapsed vs Refractory Really Means
First off, lets clear up the jargon. Relapsed AML means the disease popped up again after you had responded to a previous treatment and had entered remission. Refractory AML, on the other hand, is when the leukemia never really bows out, despite multiple rounds of therapy. Think of it as a stubborn weed that refuses to diesometimes you need a different herbicide.
Quick Comparison
| Feature | Relapsed AML | Refractory AML |
|---|---|---|
| Timing | Returns after remission | Never achieves remission |
| Typical Triggers | Residual disease, clonal evolution | Intrinsic resistance |
| Treatment Focus | Salvage therapy, transplant | Intensified or novel agents |
Why AML Sometimes Comes Back
Leukemia cells are crafty. Even after treatment, a tiny pool of minimal residual disease (MRD) can linger, hidden from standard tests. Over time, these cells can acquire new mutationslike a plot twist in a thrillerthat make them resistant to the drugs that once worked. Studies in show that clonal evolution is a major driver of relapse, especially in patients with highrisk cytogenetics.
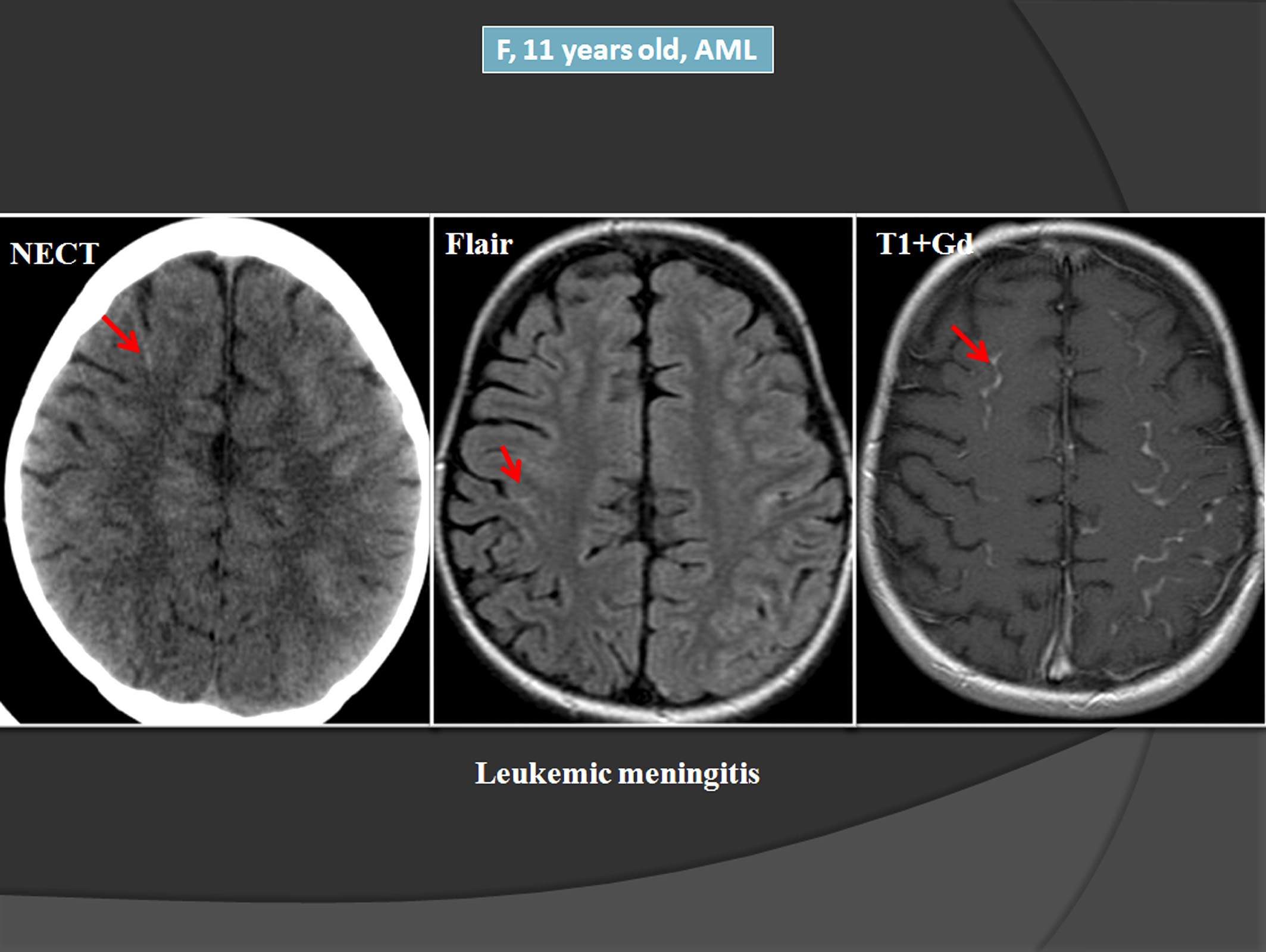
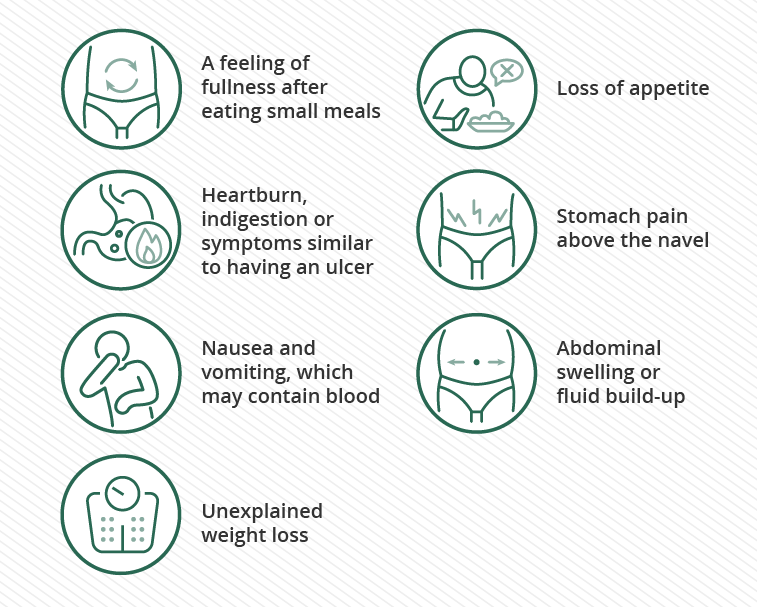
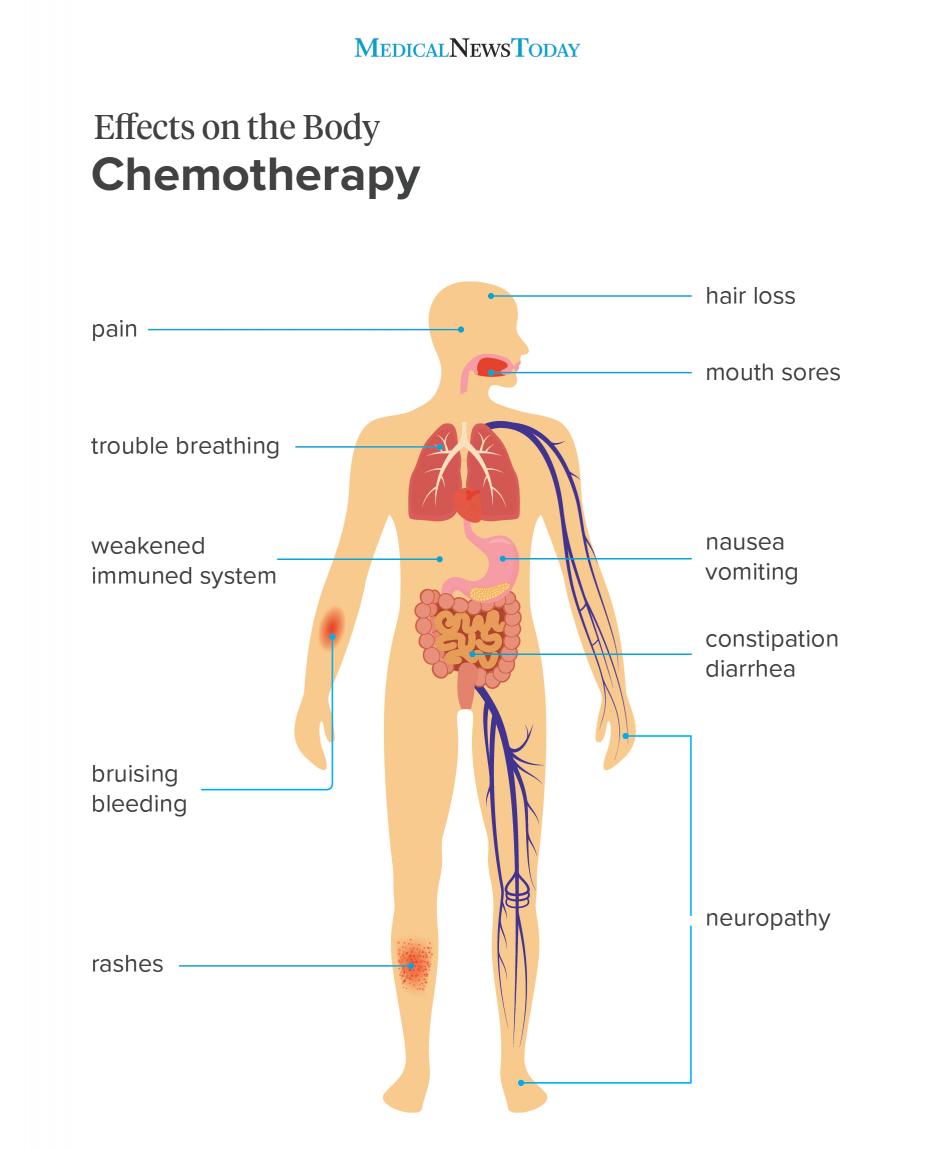
Spotting Relapse Symptoms Early
Early warning signs can feel vague, but theyre worth paying attention to:
- Sudden fatigue that doesnt improve with rest
- Unexplained bruising or bleeding
- Fever or frequent infections
- Bone pain or stiffness
- New abnormalities in blood counts
If any of these pop up, call your hematologist right away. Prompt MRD testing can make the difference between a manageable salvage regimen and a fullblown crisis.
Treatment Options
FirstLine Choices After Relapse
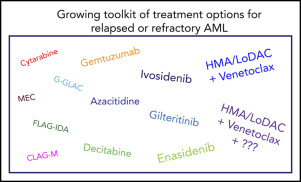
When a relapse is confirmed, doctors first assess how fit you are. Younger patients with good organ function often receive intensive chemotherapy regimens like FLAGIDA (fludarabine, cytarabine, GCSF, and idarubicin) or MEC (mitoxantrone, etoposide, cytarabine). These aim to achieve a second remission, paving the way for a possible transplant.
If youre older or have comorbidities, the approach shifts to lowerintensity options. Hypomethylating agents (HMAs) like azacitidine or decitabine, sometimes paired with the BCL2 inhibitor venetoclax, have become a mainstay. A recent metaanalysis showed median overall survival of about 79 months with HMA+venetoclax in relapsed patients, notably better than HMA alone.
Targeted and Novel Therapies
Precision medicine has changed the game for many relapsed cases. If your leukemia carries an FLT3 mutation, gilteritinib is FDAapproved and has demonstrated impressive response ratesup to 40% in some trials. Other actionable mutations like IDH1/2 can be tackled with specific inhibitors (enasidenib for IDH2, ivosidenib for IDH1).
Pros&Cons at a Glance
| Agent | Response Rate | Key Side Effects | Typical Use |
|---|---|---|---|
| Gilteritinib | 40% | QT prolongation, liver enzyme rise | FLT3mutated relapse |
| Idh Inhibitors | 30% | Differentiation syndrome | IDHmutated relapse |
| Venetoclax+HMA | 3545% | Neutropenia, infections | Older/frail patients |
Allogeneic StemCell Transplant (SCT)
If youre a candidate, an allogeneic SCT remains the only potentially curative option. The graftversusleukemia effect can eradicate lingering leukemic cells that survived chemotherapy. According to the , transplant is recommended for eligible patients after achieving a second remission, or even as a bridging therapy in certain highrisk relapses.
Even after a transplant, relapse can occur. In those cases, options include donorlymphocyte infusions, azacitidine maintenance, or enrolment in a clinical trial exploring posttransplant targeted agents.
Clinical Trials: The Frontier
When standard care feels like a deadend, clinical trials can offer access to cuttingedge drugsthink nextgen FLT3 inhibitors, bispecific antibodies, or CART cell therapies. Ask your doctor about ongoing studies, or search clinicaltrials.gov with relapsed AML as a keyword. Even if you dont qualify, the trial registry can give you a sense of where the field is heading.
Prognosis & Life Expectancy
What the Numbers Say
Lets talk raw data. Median overall survival (OS) after relapse varies dramatically based on treatment intensity:
- Intensive salvage chemo without transplant: ~46 months
- HMA+venetoclax: 79 months
- Targeted agents (e.g., gilteritinib) in mutationpositive disease: 1012 months
- Allogeneic SCT (when successful): >1224 months, with a subset achieving longterm remission.
These figures arent destinyindividual factors like age, cytogenetics, MRD status, and how quickly you start salvage therapy can shift the odds.
Key Factors Shaping Prognosis
Consider the following variables when you or a loved one hears the word prognosis:
- Age: Younger patients generally tolerate aggressive regimens better.
- Cytogenetics & Mutations: Complex karyotype or TP53 mutations correlate with poorer outcomes.
- Time to Relapse: Relapse within 6 months of initial remission signals a more aggressive disease.
- MRD Negativity: Achieving MRDnegative status before transplant dramatically improves survival.
Quality of Life Matters
Survival statistics are important, but theyre only part of the story. Side effects like prolonged neutropenia, mucositis, or organ toxicity can impact daily living. Many patients and families now weigh qualityadjusted life years (QALYs) alongside raw survival time. Supportive caregrowth factors, infection prophylaxis, nutrition counselingcan make a huge difference in staying comfortable while fighting the disease.
Managing Risks & Supporting Recovery
Handling Toxicities HeadOn
Intensive chemotherapy can feel like walking a tightrope. Here are a few practical tips:
- Neutropenia: Keep a fever log, practice strict hand hygiene, and have a low threshold for calling your care team.
- Organ Monitoring: Regular liver and kidney panels catch early signs of druginduced injury.
- Nutrition: Small, frequent meals with proteinrich foods help maintain strength during cytopenic phases.
Preventing Another Relapse
Even after youve conquered a relapse, vigilance remains key. Strategies include:
- Routine MRD assessments every 36 months (bone marrow or peripheral blood).
- Maintenance therapylowdose azacitidine or oral FLT3 inhibitorshas shown promise in extending remission.
- Staying on schedule with followup appointments and lab work.
For patients concerned about longterm survival and outlook after aggressive interventions, reading about prostate cancer outlook can sometimes help frame expectations about qualityoflife tradeoffs and survivorship planning across different cancer types.
Lifestyle & Emotional Support
Science tells us that a balanced diet, gentle exercise (think walking or yoga), and stressreduction techniques can boost overall resilience. But perhaps more critical is a solid emotional support network. Whether its a patientadvocacy group, a close friend who listens, or a therapist specializing in oncology, sharing fears and triumphs can lighten the mental load.
RealWorld Stories
Case Study A: FLT3Mutated Relapse, Gilteritinib Success
John, 58, relapsed six months after his first remission. Genetic testing revealed an FLT3ITD mutation. He started gilteritinib and achieved MRDnegative remission within three months. He later underwent a matched sibling transplant, and two years later hes still diseasefree. His story underscores the power of targeted therapy combined with transplant when the right mutation is present.
Case Study B: PostTransplant Relapse Managed with Azacitidine + Venetoclax
Maria, 62, received an allogeneic transplant after a second remission. Six months later, a subtle rise in blasts suggested early relapse. Rather than aggressive chemo, her team chose lowdose azacitidine plus venetoclax. She tolerated the regimen well, achieved another remission, and remains on maintenance therapy, enjoying gardening and weekend hikes.
Patient Voice: I Thought My Journey Was Over
When my doctor said relapse, my heart stopped. But I wasnt ready to surrender. The support from my oncology nurse, the clear explanations about each treatment option, and the fact that I could still choose a path that fit my lifethose made all the difference. Anonymous survivor, 2023.
Conclusion
Facing a relapse of AML is undeniably tough, but youre not wandering this road alone. By recognizing symptoms early, understanding the full spectrum of relapsed AML treatment protocols, and weighing the realistic expectations for life expectancy, you can make informed decisions that align with both your health goals and personal values. Remember, every piece of datawhether its a response rate or a sideeffect profileserves as a compass, guiding you toward the safest, most effective route.
Take the next step: talk openly with your hematologist about your options, ask about clinical trials that might fit your situation, and lean on trusted support networks. With the right blend of medical expertise, personal courage, and practical care, you can navigate this chapter with confidence and hope.
FAQs
What does relapsed AML mean?
Relapsed AML means leukemia has returned after initially responding to treatment and entering remission, indicating the disease has come back.
What are common treatment options for relapsed AML?
Treatment may include intensive chemotherapy regimens like FLAG-IDA or MEC, hypomethylating agents with venetoclax for older patients, targeted therapies such as gilteritinib for FLT3 mutations, and potentially an allogeneic stem cell transplant.
How important is a stem cell transplant in relapsed AML?
Allogeneic stem cell transplant is currently the only potentially curative option for eligible patients after achieving a second remission and can improve long-term survival.
What factors influence prognosis after AML relapse?
Prognosis depends on age, genetic mutations or cytogenetics, time to relapse, minimal residual disease status, and how quickly salvage therapy begins.
Are there clinical trials available for relapsed AML patients?
Yes, clinical trials offer access to new treatments like next-generation FLT3 inhibitors, bispecific antibodies, and CAR-T therapies, which may benefit patients with limited standard options.