Familial dysautonomia (FD) is a rare inherited disorder caused by mutations in the ELP1/IKBKAP gene. Knowing the genetics behind FD tells you why it runs in families, who might be a carrier, and what testing options are available.
Lets walk through the science, the testing process, and what the genetics mean for everyday lifewithout the jargon. Think of this as a friendly chat with someone whos been there, ready to share the most helpful information.
What Is FD?
Definition and Core Features
FD, also called Riley-Day syndrome, is a disorder of the autonomic nervous system. It affects how the body controls blood pressure, temperature, digestion, and many other automatic functions. People with FD often experience frequent dizziness, feeding difficulties, and reduced pain sensation.
FD vs. General Dysautonomia
| Aspect | Familial Dysautonomia | General Dysautonomia |
|---|---|---|
| Cause | Inherited mutation in ELP1/IKBKAP | Various causes (genetic, autoimmune, idiopathic) |
| Onset | Infancy or early childhood | Any age, often later onset |
| Inheritance | Autosomalrecessive | Usually not inherited |
| Key Symptoms | Bloodpressure swings, lack of tears, gastrointestinal issues | Heartrate variability, orthostatic intolerance |
Genetic Basis
Which Gene Triggers FD?
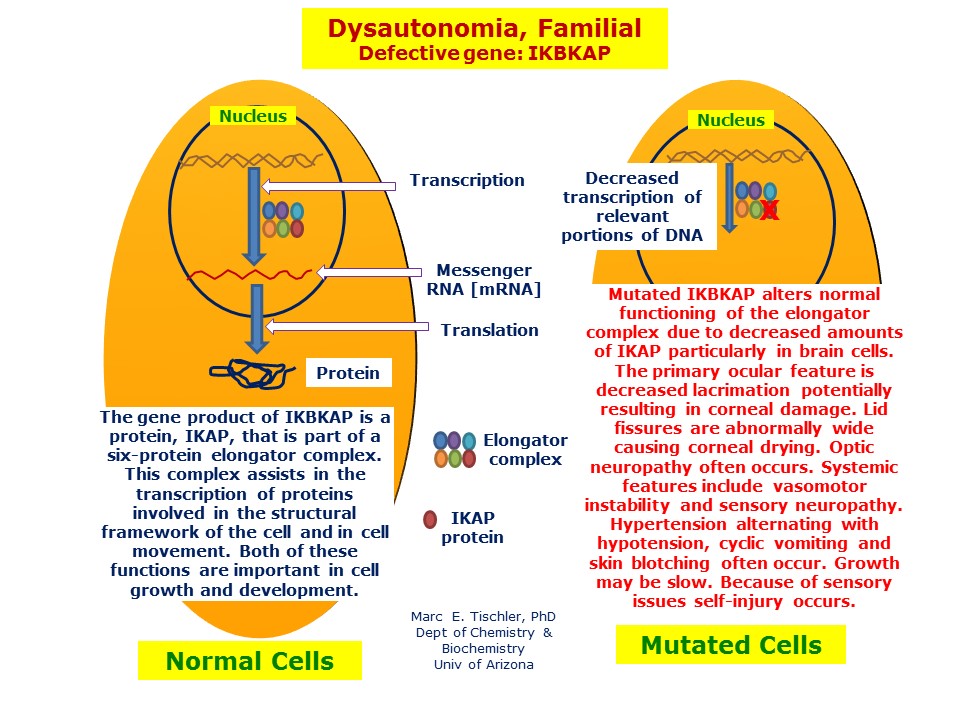
The culprit is the ELP1 genealso known as IKBKAP. It codes for a protein thats part of the Elongator complex, essential for proper nerve cell development. When the gene is faulty, the nervous system never gets the signal it needs to grow correctly.
The Common Mutation
Over 99% of FD cases share a single point mutation: c.2204+6T>C in intron20. This tiny change messes up RNA splicing, leading to dramatically reduced IKAP protein levels. The result? The autonomic nerves dont develop as they should.
Founder Effect in Ashkenazi Jews
The mutation is most prevalent among people of Ashkenazi Jewish descent. Historical migration patterns caused a founder effect, meaning the mutated allele became common in that community. Recent data from the NYU Langone Health Genetic Screening Program shows roughly 1 in 30 Ashkenazi Jews carries the mutation.
Inheritance Patterns
AutosomalRecessive Explained
FD follows an autosomalrecessive pattern. Both parents must each pass on a copy of the mutated gene for a child to be affected. If you inherit just one copy, youre a carrierbut you wont show symptoms.
Understanding Carrier Status
Being a carrier is like holding a secret keyyou can pass it to your children, but you wont feel its effects. Carrier testing is especially important for couples with a family history of FD or for those of Ashkenazi heritage. Many genetic counselors use a simple probability calculator: if both partners are carriers, theres a25% chance each pregnancy will result in an affected child.
RealWorld Example
Take Maya, who discovered she was a carrier during preconception counseling. She and her partner, both carriers, decided to pursue preimplantation genetic testing. Their story, shared in a recent case study, illustrates how knowledge of genetics turns anxiety into proactive planning.
Genetic Testing
Available Tests
Testing options include:
- Targeted DNA sequencing for the founder mutation.
- Comprehensive carrier panels that check ELP1 alongside other recessive diseases.
- Prenatal testing (amniocentesis or CVS) if a pregnancy is already underway.
When to Test?
If you have a family member diagnosed with FD, if you belong to a highrisk ethnicity, or if youre planning a family, testing can provide clarity. Newborn screening programs in certain U.S. states now include a reflex test for the FD mutation, catching cases early.
StepbyStep Guide
| Step | What to Do |
|---|---|
| 1. Choose a Lab | Select a CLIAcertified lab (e.g., Invitae, Myriad). |
| 2. Provide Sample | Send a saliva kit or a blood draw. |
| 3. Receive Results | Results classify you as positive, carrier, or negative. |
| 4. FollowUp | Discuss outcomes with a genetic counselor. |
Accuracy and Limitations
Targeted testing for the founder mutation is >99% sensitive. However, rare atypical mutations can be missed if only the common variant is screened. Thats why comprehensive sequencing is recommended when family history suggests FD but the basic test is negative.
Symptoms & Progression
Do Specific Mutations Shape Symptoms?
So far, the major founder mutation produces a fairly uniform clinical picture. Emerging research hints that modifier genesother genetic factorsmight explain why some individuals experience milder disease. A recent study in suggests that variations in the GABRB3 gene could temper autonomic instability.
Life Expectancy Trends
Decades ago, many with FD didnt survive past childhood. Thanks to multidisciplinary care, the average life expectancy now stretches into the mid40s or early50s. The Cleveland Clinic reports that early respiratory support and proactive gastrointestinal management are the biggest factors behind this improvement.
Adults Living with FD
Adulthood brings new challenges: employment, independent living, and navigating romantic relationships. Yet many adults with FD report a high quality of life when supported by a coordinated care team. Community groups, such as the FD Foundation, share practical tips on everything from cooking adaptations to accessible travel.
Treatment Options
Current SymptomFocused Care
Theres no cure yet, but treatment aims to smooth out the bumps:
- Bloodpressurestabilizing meds (midodrine, fludrocortisone).
- Feeding tubes or specialized diets for severe dysphagia.
- Physical therapy to maintain muscle tone.
- Regular ophthalmology visits to address tearproduction issues.
Experimental GeneTherapy Trials
Scientists are exploring ways to boost IKAP protein production. The NIH ClinicalTrials.gov database lists several earlyphase trials using smallmolecule readthrough agents and viralvector gene delivery. While still experimental, these studies give families hope for a future where the genetic defect can be corrected.
How Genetics Guides Management
Knowing a patients carrier status can influence family planning, while understanding the exact mutation informs eligibility for certain clinical trials. Genetic counseling becomes a cornerstone of care, helping families weigh the benefits and risks of each option.
Resources & Next Steps
Trusted Organizations
Feel free to explore these reputable groups for deeper information and community support:
- Familial Dysautonomia Foundation offers research updates and patient stories.
- National Organization for Rare Disorders (NORD) provides a concise disease handbook.
- Genetic and Rare Diseases Information Center a reliable government source.
Finding a Testing Lab
Major CLIAcertified labs like and list FD on their carrier panels. Check whether your insurance covers the test, and always request a pretest genetic counseling session.
Support Communities
Online forums (such as the FD Foundations private Facebook group) let you connect with families who truly understand the daytoday realities. Sharing experiences can turn isolation into empowerment.
Conclusion
Familial dysautonomia genetics is the key that unlocks why this rare disorder appears in families, who might be a silent carrier, and what options exist for testing and care. By understanding the ELP1/IKBKAP mutation, you gain the power to make informed decisionswhether that means pursuing genetic testing, joining a clinical trial, or simply finding the right support network.
If you or a loved one is navigating FD, reach out to a genetic counselor, explore the resources above, and remember: knowledge is a compassionate ally. What questions do you have about FD genetics? Share your thoughts or stories in the commentstogether we can turn curiosity into confidence.
For families exploring insurance and financial help for rare disease therapies, you may find additional assistance options and guidance on Exondys 51 assistance useful as an example of how support programs work for complex treatments.
FAQs
What gene is responsible for familial dysautonomia?
The disorder is caused by mutations in the ELP1 gene, also known as IKBKAP, which encodes a subunit of the Elongator complex essential for neuronal development.
How is familial dysautonomia inherited?
It follows an autosomal‑recessive pattern: a child must receive one mutated copy of ELP1 from each parent to be affected. Carriers have one normal and one mutated copy and are usually symptom‑free.
What is the most common mutation in FD patients?
More than 99 % of cases carry the founder mutation c.2204+6T>C in intron 20 of ELP1, which disrupts RNA splicing and reduces functional IKAP protein.
Who should consider genetic testing for FD?
Anyone with a family history of FD, individuals of Ashkenazi Jewish ancestry, or couples planning a pregnancy who want to assess carrier risk should discuss testing with a genetic counselor.
Are there any therapies targeting the genetic cause of FD?
Current research includes small‑molecule read‑through agents and viral‑vector gene‑therapy trials aiming to increase IKAP protein levels, though these are still experimental.