Looking for the different kinds of FDA warning letters? In the next few minutes youll see a clear list, learn why they matter, and discover how to read and act on them without getting lost in legal jargon.
From general notices to untitled closeout letters, well break down each type, show you where to find them in the FDA warning letter database, and give quick tips on what to do next. Ready? Lets dive in.
What Are Warning Letters
Definition and Legal Basis
An FDA warning letter is a formal written notice that tells a company its violating federal regulations.The agency issues it under the authority of the Federal Food, Drug, and Cosmetic Act (FD&C Act) to protect public health and push firms toward corrective action.
Why the FDA Sends Them
Think of a warning letter as a red flag from the regulator. It signals that something could endanger patients, consumers, or the environmentanything from poor manufacturing practices to misleading advertising. By handing you the letter, the FDA hopes youll fix the problem before it escalates to fines, product seizures, or even injunctions.
Finding Letters in the Official Database
The FDA maintains a searchable . You can filter by year, issuing center (e.g., CDER for drugs, CFSAN for food), or even specific keywords like pharmaceutical companies to zero in on relevant notices.
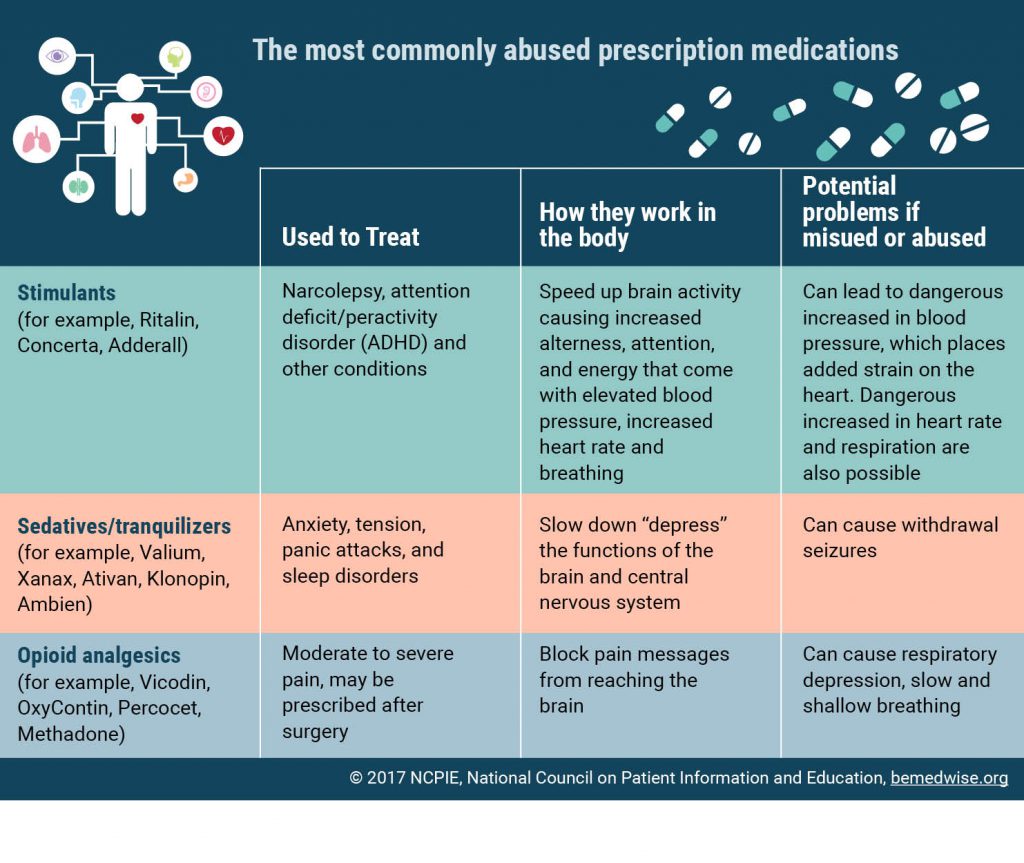
Main Types Explained
| Letter Type | Issuing Office / Scope | Typical Violations | Key Identifier |
|---|---|---|---|
| General Warning Letter | All FDA centers (CDER, CDRH, CFSAN, etc.) | CGMP failures, adulteration, misbranding | Header reads Warning Letter |
| Untitled (CloseOut) Letter | District Offices, specific centers | Minor technical issues, resolved after 483 | No Warning in title; often says CloseOut |
| 483Based Warning Letter | All centers, following a Form 483 | Same observations noted on the 483 | References Form 483 in opening paragraph |
| Tobacco Retail Letter | Center for Tobacco Products | Retail sales to minors, labeling gaps | Title includes Tobacco Retail |
| Drug Marketing Letter | CDER | False claims, offlabel promotion | Mentions Drug Marketing or Advertising |
| Food & Dietary Supplement Letter | CFSAN | Safety violations, nutrient content errors | Tagged Food or Dietary Supplement |
| Medical Device Letter | CDRH | Design control lapses, quality system gaps | Header includes Medical Device |
| OPDP Warning Letter | Office of Pharmaceutical Development & Policy | IND/ NDA deficiencies, clinical trial issues | Reference OPDP in opening line |
General FDA Warning Letters
These are the most common. They cover everything from a drug manufacturers failure to meet current good manufacturing practices (CGMP) to a food companys mislabeled ingredient list. The letter typically gives a clear description of the violation, a deadline (often 15 days) to respond, and a request for a corrective action plan.
Untitled (CloseOut) Letters
When a company quickly fixes a problem after a Form 483 inspection, the FDA may issue an untitled lettersometimes just called a closeout. Its less intimidating because it signals that the agency believes the issue is resolved, but it still serves as a record of the original noncompliance.
483Based Warning Letters
The Form 483 is the FDAs youre being observed notice. If the firm doesnt adequately address the observations, the agency escalates to a fullblown warning letter that references the original 483. Knowing that link helps you trace the audit trail and understand why the FDA is pressing for specific changes.
Specialty Letters (Tobacco, Drug Marketing, Food, Device, OPDP)
Each FDA center has its own focus. For example, a tobacco retail warning letter will zero in on sales to minors, while an OPDP warning letter deals with drug development data gaps. Recognizing the specialty tells you which regulatory experts youll need to involve. If the letter concerns drug manufacturing practices, reviewing guidance on how anti-androgens work and their safe marketing can help contextualize drug-specific concerns see an overview of how anti-androgens work for background on drug action and safety messaging.
Searching the Database
Filtering by Issue Date
If you only want the most recent insights, set the filter to 2024 or Last 30 days. That way youll see the newest FDA warning letters 2024 and can spot emerging trendslike the recent surge in dietarysupplement notices.
Keyword Searches for Targeted Results
Type specific phrases into the search bar: pharmaceutical companies, FDA 483 warning letters, or untitled letters. The engine will pull every matching document, letting you build a quick fda warning letter list for your internal audit.
Exporting and Saving for Compliance Teams
Most browsers let you save the PDF version of each letter. When youre building a compliance library, name files consistentlye.g., 2024_07_15_CDER_WL_ABCPharma.pdf. That naming convention simplifies retrieval during inspections or internal reviews.
Responding After Receipt
Immediate Response Checklist
When the envelope lands, you typically have 15 days to reply. Heres a quick checklist:
- Read the letter line by linedont skim.
- Assign a point person from regulatory affairs.
- Draft a written response acknowledging each observation.
- Outline a correctiveandpreventive action (CAPA) plan with timelines.
- Submit the response via the FDAs electronic portal (if requested).
Building a CAPA Plan
A solid CAPA has three parts: corrective actions (what youll fix now), preventive actions (how youll stop it happening again), and verification (how youll prove the fix works). Including supporting databatch records, training logs, validation reportsshows the FDA youre taking the issue seriously.
Communicating with the Agency
Use a respectful yet confident tone. Address the FDA reviewer by name (if provided), keep the language clear, and attach any evidence you reference. If youre unsure about a technical detail, consider getting input from a who has spoken to the agency before.
Legal and Reputation Considerations
A warning letter isnt a verdict, but it can affect investors, partners, and customers. Transparency is keyissue a brief public statement (if the product is already on the market) that acknowledges the issue and outlines the steps youre taking. This proactive approach mitigates reputational damage.
2024 Trends Snapshot
Volume and Hot Spots
According to the FDAs 2024 enforcement report, the number of warning letters issued to pharmaceutical firms rose 12% over 2023, driven largely by CGMP deficiencies in sterile manufacturing. At the same time, dietarysupplement letters spiked as the agency tightened scrutiny on new dietary ingredient claims.
HighProfile Cases
One notable fda warning letters pharmaceutical companies case involved a large biotech that received a warning for inadequate sterility testing on its oncology drug. The firms swift public acknowledgment and a fivestep CAPA plan earned praise from industry observers for its transparency.
Emerging Focus Areas
Artificialintelligencedriven medical devices are now appearing on the fda warning letter list. The FDA is looking for robust premarket validation and postmarket monitoringso if youre developing a device with machinelearning algorithms, start documenting your risk assessments early.
Helpful Resources
Official FDA Page
The most reliable source is the FDAs own . Its updated daily and includes PDFs of every notice.
Guidance Documents
Look for How to Respond to an FDA Warning Letter (FDA Guidance, 2022). It walks you through the response timeline, necessary content, and how to request a meeting if clarification is needed.
Industry Guides
Webinars from compliancefocused firms like GreenlightGuru often break down recent letters and share practical templates. A quick search for FDA warning letters 2024 webinar will surface several free recordings.
Legal Counsel Directories
If you need legal advice, platforms such as MartindaleHarbor list attorneys with FDA enforcement experience. Having a lawyer whos handled OPDP and CDRH cases can be a lifesaver when the stakes are high.
Conclusion
Understanding the different types of FDA warning lettersand where to find themgives you a clear roadmap to stay compliant, protect public health, and avoid costly enforcement actions. Whether youre scanning the FDA warning letter database for the latest 2024 notices or responding to an untitled closeout letter, the steps outlined above will help you act quickly and confidently. Got a specific letter youre wrestling with? Share your experience in the comments or reach out for personalized guidancewere here to help you navigate the regulatory maze.
FAQs
What is the difference between a general warning letter and an untitled (close‑out) letter?
A general warning letter flags a significant regulatory violation and requires a formal response with a corrective‑action plan. An untitled or close‑out letter is issued after a company promptly resolves the issue noted on a Form 483; it confirms compliance but does not carry the same enforcement weight.
How long do I have to respond to an FDA warning letter?
Regulators typically give you 15 calendar days to submit a written response. The reply must address each observation, provide a corrective‑and‑preventive action (CAPA) plan, and include supporting documentation.
Where can I search the FDA warning letter database for specific letters?
Use the FDA’s official portal at https://www.fda.gov/inspections‑compliance‑enforcement‑and‑criminal‑investigations/warning‑letters. Filters let you narrow by year, center (e.g., CDER, CFSAN), or keywords such as “pharmaceutical companies” or “untitled letters.”
What should be included in a corrective‑and‑preventive action (CAPA) plan after receiving a warning letter?
A solid CAPA contains: (1) corrective actions that directly fix the current violation, (2) preventive actions that stop recurrence, (3) a timeline with responsible persons, and (4) verification steps (e.g., re‑testing, audit reports) to prove effectiveness. Attach relevant records like batch logs, training sheets, and validation data.
Can a warning letter affect my company's reputation and what steps can mitigate this?
Yes. A warning letter can raise concerns among investors, partners, and customers. Mitigate impact by issuing a transparent public statement that acknowledges the issue, outlines the corrective plan, and highlights the commitment to safety. Prompt, thorough remediation also demonstrates responsibility to regulators and stakeholders.