If youve just heard the words stage4 small cell lung cancer and wonder whats actually out there right now, youre not alone. The good news is that in the last few years, several new drugs have broken through the FDAs doors, and immunotherapy combos are shifting the survival curve a bit higher. At the same time, every option comes with its own set of benefits and risks, so a balanced, doctorguided plan is essential.
Below, Ill walk you through the most recent FDA approvals, the hottest immunotherapy trials, what precision medicine looks like for SCLC, and realistic expectations for life expectancy. Think of this as a friendly chat over coffeestraight answers, a dash of empathy, and a few realworld stories to keep things grounded.
Why Stay Updated
Small cell lung cancer (SCLC) has long been known for its aggressive nature, but staying on top of the latest therapies can make a tangible difference. New approvals are not just labcoat buzz; they translate into extra weeks or months of quality time, and sometimes even a change in how sideeffects are managed. Plus, clinical trials are constantly evolving, and being aware of them lets you (or a loved one) consider a trial before it fills up.
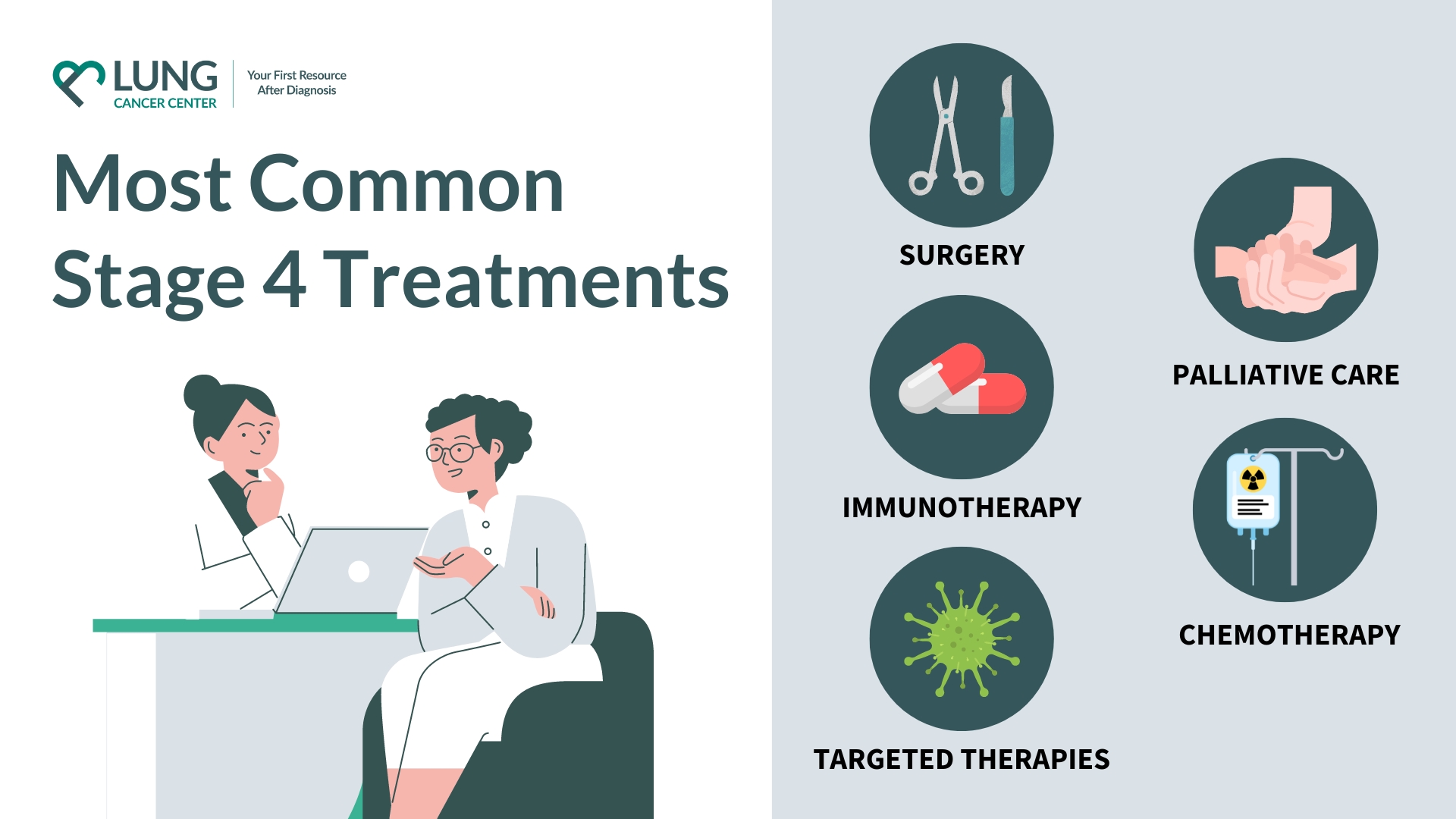
FDAApproved Drugs
Lurbinectedin (Zepzelca)
Lurbinectedin earned FDA approval in 2020 for patients whose disease has come back after platinumbased chemotherapy. It works by binding to the DNA of cancer cells, essentially throwing a wrench into their replication machinery. In the pivotal trial (NCT02566993), the median overall survival (OS) improved by about 23months compared with best supportive care.
Pros: Works where many other agents fail; modest response rate (1520%).
Cons: Can cause neutropenia, fatigue, and elevated liver enzymes. Monitoring blood counts every two weeks is usually recommended.
John, a 62yearold from Ohio, shared his experience in a video interview (linked ). After adding lurbinectedin, his survival stretched from the expected three months to ninean extra six months to spend with his grandkids.
Tarlatamab (Imdelltra)
Approved in May2024, tarlatamab is the first FDAcleared bispecific Tcell engager that targets DLL3, a protein overexpressed on most SCLC cells. By bringing Tcells into close contact with the tumor, it triggers an immune attack.
In the phaseIII trial (NCT04623204), patients receiving tarlatamab after at least one prior therapy saw a median OS gain of about 4months versus standard chemotherapy. The safety profile is generally manageable, though about 30% experienced a lowgrade cytokine release syndrome (CRS) that resolved with supportive care.
Dr. Mark Awad of Memorial Sloan Kettering notes, Tarlatamab feels like the first real leap toward a targeted immunotherapy for small cell lung cancer ().
Chemo+Atezolizumab (Tecentriq)
Since the 2022 label update, the combination of carboplatinetoposide with atezolizumab (a PDL1 checkpoint inhibitor) has become a standard firstline option for stage4 SCLC. The IMpower133 trial demonstrated an OS increase from 9.9 to 12.3months, and a modest improvement in progressionfree survival.
| Regimen | Median OS | 12mo PFS | Grade34 AEs |
|---|---|---|---|
| Chemo+Atezolizumab | 12.3mo | 5.6mo | 38% |
| Chemo alone | 9.9mo | 4.2mo | 33% |
While the numbers may look modest, many patients report a better quality of life because the immunotherapy may keep the cancer from flaring up as quickly.
Emerging Immunotherapy
Bispecific TCell Engagers Beyond Tarlatamab
Researchers are now testing a family of DLL3targeted bispecificslike AMG757 (RovaT)in earlyphase studies. Early data suggest response rates climbing into the mid20s, with manageable toxicities.
CheckpointInhibitor Combos
Combining PD1/PDL1 blockers with CTLA4 inhibitors (e.g., nivolumab+ipilimumab) has shown promise in relapsed SCLC. A 2025 JITC analysis projected a 1015% rise in twoyear survival when these combos are introduced early ().
Impact on Life Expectancy
Adding immunotherapy to chemotherapy typically adds 23months to median OS, while the newest bispecifics may push that gain to 45months. Its not a miracle cure, but every month matters.
Targeted Precision
Molecular Profiling You Should Ask For
Even though SCLC doesnt have as many actionable mutations as nonsmall cell lung cancer, a comprehensive panel (such as FoundationOne CDx) can reveal DLL3 expression, tumor mutational burden (TMB), and other markers that guide therapy choices. If DLL3 testing shows high expression, newer agents like tarlatamab may be an option worth discussing with your team; ask your oncologist whether DLL3 testing is available as part of molecular profiling.
| Molecular Marker | Targeted Option | Trial Outcome |
|---|---|---|
| DLL3 high | Tarlatamab | ORR24% |
| High TMB | Pembrolizumab | Durable responses>12mo |
Debunking the Miracle Cure Myth
Lets be clear: there is no overnight, bulletproof cure for stage4 SCLC. The hype around miracle cures often comes from unverified websites or unscrupulous supplement sellers. Trusting FDAapproved data and oncologist guidance is the safest path.
How to Spot Fake Claims
- Check if the product claims FDA approvallook for the FDA logo or an official label.
- Ask for peerreviewed study links; genuine research is usually published in journals like Lancet Oncology or JCO.
- Beware of guaranteed results or 100% cure languagecancer treatment always involves probabilities, not certainties.
Life Expectancy & Quality of Life
Current Survival Numbers
A 2025 metaanalysis of over 3,000 patients with stage4 SCLC reported a median OS of roughly 10months with any treatment, extending to about 14months when chemo+atezolizumab is used.
Longest Survivor Stories
Sarah, a 48yearold nonsmoker, entered a clinical trial combining tarlatamab with radiation in 2023. Five years later, shes still alive and actively advocating for patientdriven research. Her story, featured in the 2024 ASCO meeting, underscores that outliers exist, especially when trials are entered early ().
Factors That Stretch Survival
| Factor | Typical OS Gain |
|---|---|
| Early trial enrollment | +35mo |
| Good performance status (ECOG01) | +23mo |
| Comprehensive supportive care (nutrition, palliative services) | +12mo |
Managing SideEffects
Sideeffects are part of the journey, but they dont have to dominate it. Here are a few tricks Ive heard from patients and oncology nurses:
- Antiemetics: A combination of ondansetron and dexamethasone often keeps nausea at bay.
- Growthfactor support: Filgrastim (GCSF) can reduce the depth and duration of neutropenia.
- Fatigue combat: Light activity (short walks), scheduled rest, and a balanced diet rich in protein help keep energy levels steadier.
- Immunerelated AEs: If you develop rash, diarrhea, or shortness of breath, contact your team earlycorticosteroids can quickly reverse many of these issues.
Talking to Your Oncologist
Preparing a Concise Agenda
Bring a fresh copy of your latest scan, a list of current meds, and written questions. Example starter: I read about tarlatamab and wonder if my tumors DLL3 expression makes me a candidate.
When to Consider a Clinical Trial
Eligibility usually hinges on performance status (ECOG02), prior lines of therapy, and sometimes specific biomarkers. The National Cancer Institutes trial finder and the Lung Cancer Research Foundations patientmatch service are helpful tools ().
Supporting Resources
- Patientadvocacy groups like the Lung Cancer Foundation of America (LCFA) offer webinars on navigating new therapies.
- The Cancer Research Institute provides a free Ask an Expert session each monthgreat for clarifying doubts about immunotherapy.
Conclusion
Here are the three takeaways you can act on right now:
- New FDAapproved optionslurbinectedin, tarlatamab, and chemo+atezolizumabare reshaping the treatment landscape for stage4 small cell lung cancer.
- Immunotherapy combos, especially bispecific Tcell engagers, are extending median survival and are the fastestgrowing area of research.
- Partner with your oncologistrequest molecular profiling, discuss trial eligibility, and manage sideeffects proactively to make the most of every treatment month.
If youve been through any of these therapies or have questions about the next steps, Id love to hear your story in the comments. Sharing helps us all feel less alone on this tough road. And remember, youre not navigating this alonetheres a whole community of patients, doctors, and researchers working together to bring better days ahead.
FAQs
What are the newest FDA‑approved drugs for stage 4 small cell lung cancer?
Lurbinectedin (Zepzelca) and tarlatamab (Imdelltra) received FDA approval in 2020 and 2024 respectively, and the chemo + atezolizumab combo became a standard first‑line option in 2022.
How does tarlatamab work and who can receive it?
Tarlatamab is a bispecific T‑cell engager that binds DLL3 on SCLC cells and CD3 on T‑cells, directing an immune attack. It is approved for patients whose disease has progressed after at least one prior therapy and who have DLL3‑positive tumors.
Can immunotherapy improve survival for stage 4 SCLC patients?
Yes. Adding checkpoint inhibitors such as atezolizumab to chemotherapy adds about 2–3 months to median overall survival, while newer bispecific agents may extend survival by 4–5 months.
What molecular tests should I ask my doctor about?
Request a comprehensive tumor‑profiling panel (e.g., FoundationOne CDx) to assess DLL3 expression, tumor mutational burden, and any actionable mutations that could guide targeted or immunotherapy options.
When should I consider enrolling in a clinical trial?
Early enrollment is best, especially if you have good performance status (ECOG 0‑1) and have exhausted standard therapies. Use resources like the NCI trial finder or lung‑cancer foundations to locate active studies.