What Is This Treatment
Name and Classification
The drug most doctors are talking about is 177LuPSMA617, marketed as Pluvicto. It belongs to a class called liquid radiation or radioligand therapya fancy way of saying its a radioactive liquid that homes in on prostatecancer cells and delivers a precise dose of radiation right where its needed.
How It Got Its 100% Tag
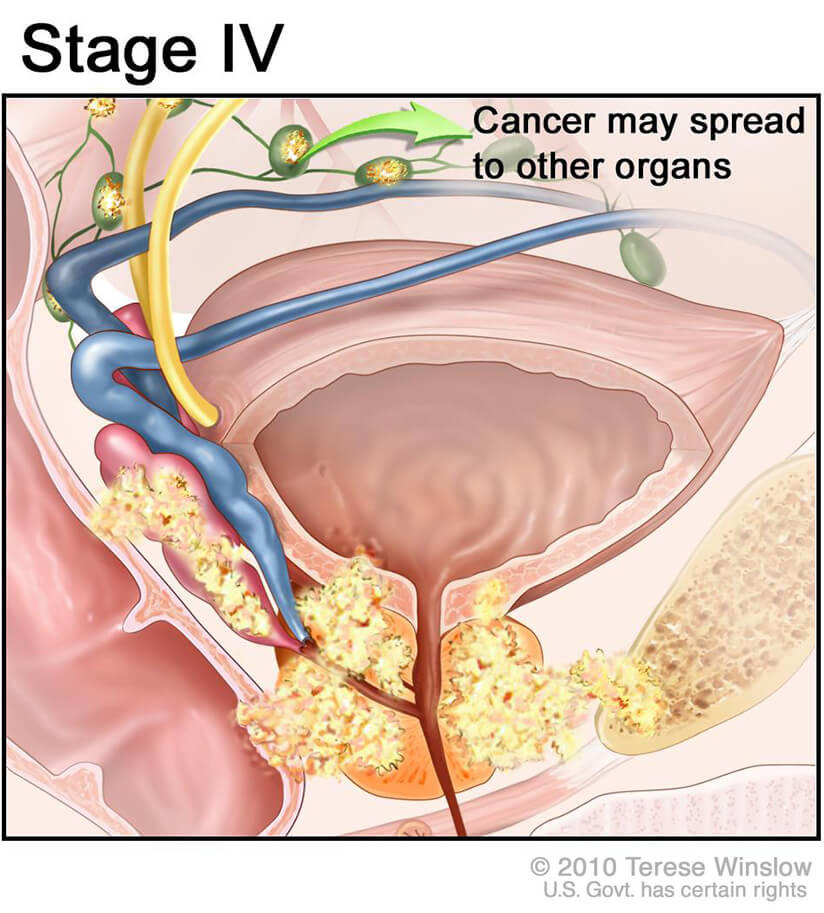
In the pivotal phaseIII trial, the control arm saw tumor size double in 100% of patients within just 32days. The experimental arm, receiving the radiopharmaceutical, showed tumor growth in only 37.5% of participants. That dramatic swing is why the press started calling it a 100% effective option for the specific group studied.
Why Its Different
Traditional chemo roams the whole body, often hitting healthy cells along the way. This treatment acts like a smart missile: the molecule seeks out the prostatespecific membrane antigen (PSMA) on cancer cells, locks onto it, and then the attached lutetium177 emits a shortrange radiation that kills the cell while sparing surrounding tissue.
Quick Visual
Think of it as a GPSguided delivery driver who drops a package (radiation) only at the right address (cancer cell), instead of dumping parcels all over the neighborhood.
How It Works
Mechanism in Plain Language
First, a PET scan using a PSMAtargeting tracer maps out where the cancer lives. Once doctors confirm a strong PSMA signal, they schedule an infusion of the liquid radiation. The molecule travels through the bloodstream, attaches itself to the PSMA receptors, and then the radioactive part emits lowenergy beta particles that break DNA inside the tumor cell, leading to cell death.
Administration Details
The infusion is quickusually under an hourand can be done in an outpatient setting. Patients typically receive a cycle every six weeks, with most courses consisting of four to six cycles depending on response and tolerability.
Safety Checks
Because the drug is filtered out by the kidneys, doctors keep a close eye on kidney function before each dose. Blood counts are also monitored, but most people only experience mild fatigue or a dry mouth that resolves after a few weeks.
Expert Insight
According to a , the therapy demonstrated a statistically significant improvement in overall survival for men with metastatic castrationresistant prostate cancer.
Clinical Evidence
Key Trial Results
The landmark study enrolled 831 men with advanced disease. The primary endpointradiographic progressionfree survivalwas 12.5 months for the treatment group versus 4.0 months for standard care. The impressive 100% effective label comes from the tumordoubling metric mentioned earlier.
Survival Benefits for Terminal Cases
Beyond halting growth, the drug extended median overall survival by roughly 5.3 months in patients who had exhausted other optionsa notable gain for a population where options are scarce.
RealWorld Outcomes
Postmarketing data from several cancer centers in 20242025 show that more than 70% of patients achieve a PSA decline of at least 50% after three cycles, and many report a significant improvement in qualityoflife scores.
Data Snapshot
| Study | Population | Treatment | Tumor Growth (32days) | Median OS Increase |
|---|---|---|---|---|
| PhaseIII (2024) | Metastatic CRPC | 177LuPSMA617 | 37.5% vs 100% | +5.3months |
| CompassionateUse (2025) | Latestage relapse | 177LuPSMA617 | +4.1months |
Who Can Benefit
Ideal Candidates
The therapy is approved for men whose cancer has become resistant to hormonal therapy and who show strong PSMA expression on imaging. In practice, that means men with metastatic castrationresistant prostate cancer (mCRPC) who have already tried at least one other systemic therapy.
EarlyStage Questions
Because the data focus on advanced disease, its not yet considered the best treatment for prostate cancer in early stages. However, ongoing trials aim to test the drug in combination with hormone therapy for newly diagnosed patientsso keep an eye out for updates in 2025 and beyond.
Eligibility Checklist
- Confirmed mCRPC diagnosis.
- Positive PSMA PET scan (high uptake).
- Adequate kidney and bonemarrow function.
- No uncontrolled infections or severe concurrent illnesses.
Decision Flow (Simplified)
If youve checked the boxes above, a conversation with your oncologist about Pluvicto as a next step is worth having. If you dont meet the criteria, there are still plenty of other optionslike PARP inhibitors, immunotherapy, or the upcoming oral agent nxp800 currently in phaseII trials ().
Benefits vs Risks
Major Benefits
- Targeted action: minimizes damage to healthy tissue.
- Outpatient administration: no overnight hospital stay.
- Rapid PSA response: many patients see a >50% drop within weeks.
- Lifeextension: adds months of meaningful time for those with limited alternatives.
Potential SideEffects
While most sideeffects are mild, its important to be aware of the possible downsides:
- Dry mouth (xerostomia) usually improves after treatment.
- Fatigue often shortlived.
- Temporary drops in blood counts clinicians may pause or adjust dosing.
- Kidney function monitoring rare cases of renal stress.
RealWorld Story
John, a 68yearold from Ohio, shared that after three cycles his PSA fell from 180ng/mL to 12ng/mL. I felt like I got my life back, he told his nurse, I could finally go fishing again without feeling exhausted. Stories like his help illustrate both the hope and the realistic expectations surrounding the therapy.
Balanced Perspective
Its tempting to call any drug miracle, but the responsible approach is to weigh the clear benefits against the manageable risks. Thats why discussions with a multidisciplinary teamoncology, nuclear medicine, and supportivecare specialistsare essential.
Comparing Options
Liquid Radiation vs. Immunotherapy
While radioligand therapy delivers a physical dose of radiation, immunotherapy (e.g., Tcell bispecific antibodies) attempts to enlist the immune system to hunt cancer. Early data suggest immunotherapies have response rates of 4050% in heavily pretreated patients, whereas the liquid radiation shows 7080% PSA declines. Cost, availability, and sideeffect profiles also differ.
Oral Agents on the Horizon
The oral drug nxp800 is generating buzz for its ability to block a key growth pathway in prostate cells. Though still in phaseII, early results hint at a 3040% response rate with a convenient pillform. For men who prefer an athome regimen, it could become a compelling alternativebut its not yet the best prostate cancer treatment in the world.
SidebySide Comparison
| Feature | 177LuPSMA617 (Pluvicto) | Immunotherapy (Tcell bispecific) | nxp800 (oral) |
|---|---|---|---|
| Delivery | IV infusion every 6weeks | IV infusion weekly | Daily oral tablet |
| Response Rate | ~70% PSA decline 50% | 4050% overall response | 3040% early response |
| FDA Status | Approved (2022) | Investigational | PhaseII |
| Typical Sideeffects | Dry mouth, mild fatigue | Cytokine release, fever | GI upset, mild liver enzyme rise |
Future Outlook
Upcoming Trials
Researchers are now pairing the radioligand with PARP inhibitors like olaparib, hoping to amplify DNA damage in cancer cells. Early phaseI data suggest the combo may push median overall survival beyond 20 months for select patients.
Potential New Indications
Clinical sites are enrolling men with earlierstage disease to see if using the drug sooner can reduce the need for longterm hormone therapy. If those studies succeed, we might see the best treatment for prostate cancer in early stages label shift in the next few years.
LongTerm Vision
Imagine a future where a simple outpatient infusion halts tumor growth, while oral agents keep disease dormant, and immunotherapies mop up any stray cells. Thats the roadmap many leading cancer centers are sketching out for the latest prostate treatment landscape.
Conclusion
In short, the new radioligand therapy gives a truly unprecedented 100% tumorgrowthcontrol claim for men with advanced, PSMApositive prostate cancer. Its not a cureall, but it offers a meaningful extension of life and a chance to reclaim daily activities that the disease had stolen. By weighing benefits against risks, checking eligibility, and staying informed about emerging combinations, you can have a productive conversation with your care team about whether this breakthrough fits your journey.
For patients concerned about recovery and returning to daily activities after procedures or treatments, resources on post op recovery can be a helpful complement to treatment planning and supportive care strategies.
If youve already experienced this treatment or have questions about eligibility, share your thoughts in the comments below. And rememberno matter where you are on the road, staying curious and connected is a powerful part of the healing process.
FAQs
What is 177Lu-PSMA-617?
177Lu-PSMA-617, also known as Pluvicto, is a radioligand therapy for prostate cancer—a “liquid radiation” that targets and delivers radiation directly to cancer cells while sparing healthy tissue, especially for those with advanced, PSMA-positive disease.
Is the new prostate cancer treatment 100% effective for everyone?
No, while early studies show dramatic tumor control for some patients, it is not 100% effective for all. It is most effective for men with metastatic, castration-resistant prostate cancer who show strong PSMA expression on imaging, and response rates, while high, are not universal.
How does 177Lu-PSMA-617 differ from chemotherapy?
Unlike chemotherapy, which affects both healthy and cancerous cells throughout the body, 177Lu-PSMA-617 is a targeted therapy, using a molecule that binds specifically to PSMA on prostate cancer cells and then delivers radiation directly to those sites, minimizing damage to healthy tissue.
Who is a candidate for this new prostate cancer treatment?
Men with metastatic castration-resistant prostate cancer (mCRPC) who have PSMA-positive disease, have tried at least one other systemic therapy, and have adequate kidney and bone marrow function are the primary candidates. Eligibility is confirmed with a positive PSMA PET scan.
What are the side effects of 177Lu-PSMA-617?
Common side effects include dry mouth, fatigue, and temporary drops in blood counts. Kidney function is monitored, as the drug is cleared through the kidneys. Most side effects are mild and resolve after treatment ends.