What Is a Warning?
Definition What does the FDA call a warning letter?
In plain English, a warning letter is the FDAs formal way of saying, Hey, youre not playing by the rules, and we need you to fix it fast. According to the , a letter is issued after an inspection when the agency finds a significant violation of the Food, Drug, and Cosmetic (FD&C) Act. Its public, its documented, and its meant to protect public health.
Why the FDA Issues Warning Letters
The agencys mission is to keep us safefrom the cereal in our pantry to the insulin we depend on. When manufacturers slip up, a warning letter nudges them (or sometimes shoves) to correct the problem before it becomes a bigger risk. Think of it as a safety net that catches issues early, saving lives and money.
Quick Comparison Warning Letter vs. FDA483 vs. Notice of Violation
| Document | Purpose | Typical Content | Next Steps |
|---|---|---|---|
| Warning Letter | Formal enforcement | Violation details, CFR citations, correctiveaction deadline | Company must respond within 15 days |
| FDA483 | Observation list | Observations made during inspection, not yet formalized | Company may address informally; may evolve into a warning letter |
| Notice of Violation | Legal notice | Specific legal breach, potential for statutory penalties | Possible civil or criminal action |
Finding the Warning List
The FDA Warning Letter Database How to Access It
Start at the FDAs . Youll see a simple search bar, filter dropdowns (Office, Date Range, Product Category), and a Search button. Its as easy as looking up a recipe, except the ingredients are compliance details.
Alternative Sources (when the FDA site is slow or down)
- Pharmaguidelines public archive (rehosts FDA letters for quick access).
- Redica Systems compliance tracker a subscription service but often offers a free preview of recent letters.
- AFDOs FDA Letters page curated for dietarysupplement companies.
Building Your Own Local Archive
If youre a compliance officer or a curious analyst, consider creating a spreadsheet. Columns you might include:
- Issuing Office
- Letter Date
- Company Name
- Product(s) Involved
- CFR Citations
- Key Violations
- Response Deadline
- Followup Outcome
Having a personal archive lets you run custom filters, spot trends, and even produce a quarterly report for your team.
Types of Warning Letters
General Warning Letters
These cover most categoriesdrugs, foods, cosmetics, and devices. They usually focus on labeling errors, manufacturing deficiencies, or unapproved claims.
Tobacco Retailer Warning Letters
Targeted at retailers who violate advertising or salestominor rules. The language is often more stringent because of the publichealth stakes.
HealthFraud & Misbranding Letters
When a company markets a product with unproven benefits, the FDA steps in. The letters cite specific FTC and FDA statutes, making them a useful case study for marketers.
FDA483Based Warning Letters
Sometimes an inspection results in a Form483 first. If the company doesnt adequately respond, the FDA escalates to a formal warning letter. This hybrid type tells you the issue was serious enough to move beyond observations.
Visual Cheat Sheet (Idea)
Picture an infographic that stacks each type, the typical trigger (e.g., missing lot number), and a headline example. You could even create one in PowerPoint for internal training.
Search the List Efficiently
Using BuiltIn Filters (Office, Date Range, Product Category)
The database lets you narrow down by:
- Office (e.g., Center for Drug Evaluation and Research)
- Date (Last 7Days, 30Days, 60Days)
- Category (Pharmaceuticals, Food, Medical Devices)
Set the range you need, click Search, and watch the list populate instantly.
Keyword Strategies for Precise Results
Combine the main keyword with specificsbrand names, NDC numbers, or terms like cGMP. Example: Pfizer FDA warning letter 2024 cGMP will slice right to the relevant entry. If youre tracking safety concerns tied to a therapy or class of drugs, add clinical termse.g., searching for anti-androgen side effects can help you find letters or safety notices that mention adverse events associated with those therapies.
Exporting Data for Analysis
While the FDA site doesnt offer a direct CSV download, you can:
- Copy the table into Excel.
- Use a browser extension such as Table Capture to export as CSV.
- Write a simple Python script with
requestsandBeautifulSoupfor automated scraping (respect the sites robots.txt!).
Sample Search WalkThrough
Lets say you want the latest letters for pharmaceutical companies. Select Pharmaceuticals under Product Category, set the date range to Last 30Days, and type pharmaceutical in the keyword box. The results will list every recent warning, complete with a PDF link.
Common Pitfalls & How to Avoid Them
- Duplicate listings: Some letters appear under multiple offices; deduplicate in your spreadsheet.
- Outdated PDFs: Occasionally a PDF is removed; use the Wayback Machine to retrieve older versions.
- Missing data: Not every letter includes a full CFR citation; crossreference with the for clarification.
Recent Letters & Trends
Top 5 Industries Receiving Letters in the Last 30 Days
- Dietary Supplements labeling and newingredient claims.
- Medical Devices software validation gaps.
- Pharmaceuticals sterility and cGMP deviations.
- Food Production undeclared allergens.
- Tobacco Retail underage sales violations.
Emerging Issues (AIdriven Devices, Novel Food Additives)
The 2024 surge in AIenabled diagnostic tools has triggered a wave of letters about algorithm transparency and data integrity. Similarly, novel food additives derived from insects are now landing on the radar, with letters focusing on safety data gaps.
Trend Chart (Idea)
Imagine a line graph showing the monthly count of warning letters over the past 12months. Youd see spikes after major FDA inspections and dips during holiday shutdownsgreat for a quarterly compliance review.
How Companies Are Responding
Many firms are adopting rapid response teams that draft correctiveaction plans (CAPs) within 24hours of receipt. A recent case study from shows a biotech company reducing its response time from 14days to 3days, ultimately avoiding a civil penalty.
Interpret & Respond Steps
Decoding the Violations Section
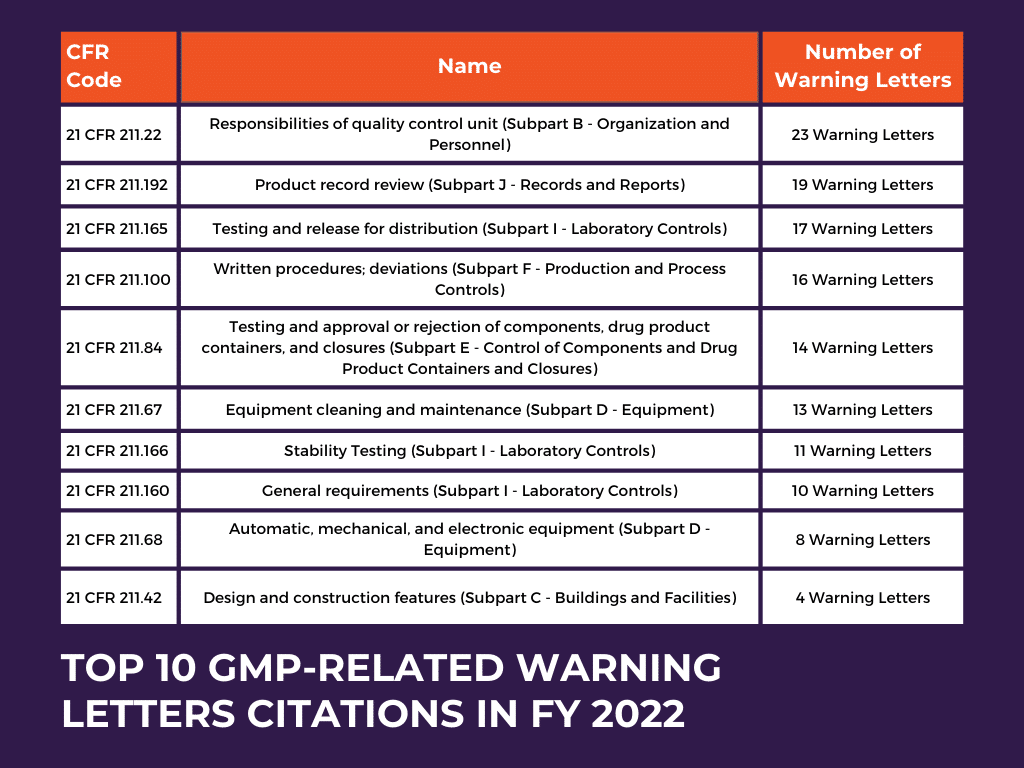
Each letter cites a Code of Federal Regulations (CFR) provisione.g., 21CFR210.3(b) for Current Good Manufacturing Practice (cGMP). If youre unfamiliar, a quick search on the FDAs CFR website will pull up the exact rule language. Understanding the legal jargon is half the battle.
5Step Response Playbook
- Acknowledge receipt: Send a written acknowledgment within 2days.
- Conduct rootcause analysis: Use a fishbone diagram to trace the issue.
- Draft correctiveaction plan (CAP): Outline what was wrong, how youll fix it, and preventive measures.
- Submit response: Meet the 15day deadline (or request an extension with a solid justification).
- Followup inspection: Prepare for a reinspection; keep all documentation handy.
Sample CAP Outline (Downloadable Template)
If youd like a starter template, think of headings like Problem Statement, RootCause, Corrective Action, Preventive Action, and Verification. Fill in details and youre ready to go.
RealWorld Example
In 2024, a midsize pharmaceutical firm received a warning letter for incomplete batch records. By applying the playbook, they corrected the documentation process, instituted electronic batch tracking, and the FDA lifted the observation at a followup visit. The story is detailed on the FDAs .
Benefits vs Risks
Benefits
- Market intelligence see where competitors stumble.
- Compliance benchmarking compare your SOPs against realworld findings.
- Consumer safety stay ahead of products that might affect public health.
Risks
- Misinterpretation taking a technical citation out of context can lead to panic.
- Legal exposure publishing confidential details without verification may breach regulations.
- Outdated info older letters might have been resolved; always check the latest status.
Checklist for Safe Use
- Verify the letters date and status (open or closed).
- Crossreference the CFR citation with the current Code.
- Document your source (URL, access date).
- Use the data for internal insights, not public speculation.
Quick FAQ Answers
Can I search the list for a specific company name?
Yesjust type the companys exact legal name into the keyword box and hit Search. The filter will return any letters issued to that entity.
Are warning letters public record?
Absolutely. The FDA publishes them in the FDA warning letter database as part of its transparency mandate.
Whats the difference between a warning letter and an FDA483?
An FDA483 is an observation list given at the end of an inspection, while a warning letter is a formal enforcement action that follows if the issues arent corrected.
How often is the database updated?
Typically within 2448hours of issuance. Youll see a Date Published stamp on each entry.
Conclusion
There you have ityour friendly guide to the FDA warning letter list. From understanding what a warning letter really means, to mastering the search tools, spotting trends, and crafting a solid response, you now have a roadmap that turns raw regulatory data into actionable insight. Bookmark the official FDA warning letters page, set a quarterly reminder to scan new entries, and consider building that personal archive we talked about. If you have questions, experiences, or even a favorite compliance tip, drop a comment belowlets keep the conversation going. Together, we can stay informed, stay compliant, and keep the public safe.
FAQs
How can I access the FDA warning letter list?
Visit the official FDA warning letter database at fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters, where you can search by office, date range, product category, or keyword.
What information is included in a warning letter?
Each letter provides the issuing office, date, company name, product(s) involved, specific CFR citations, a detailed description of the violations, a corrective‑action deadline, and any follow‑up outcome.
How often is the FDA warning letter database updated?
The database is refreshed within 24–48 hours of a letter’s issuance, and each entry shows a “Date Published” stamp so you know it’s current.
What’s the difference between an FDA 483 and a warning letter?
An FDA 483 lists observations made during an inspection and is not a formal enforcement action. If the company fails to adequately address the observations, the FDA may issue a formal warning letter that requires a corrective‑action plan.
What steps should a company take after receiving a warning letter?
Follow the 5‑step playbook: acknowledge receipt within 2 days, conduct a root‑cause analysis, draft a corrective‑action plan, submit the response within the 15‑day deadline, and prepare for the follow‑up inspection.