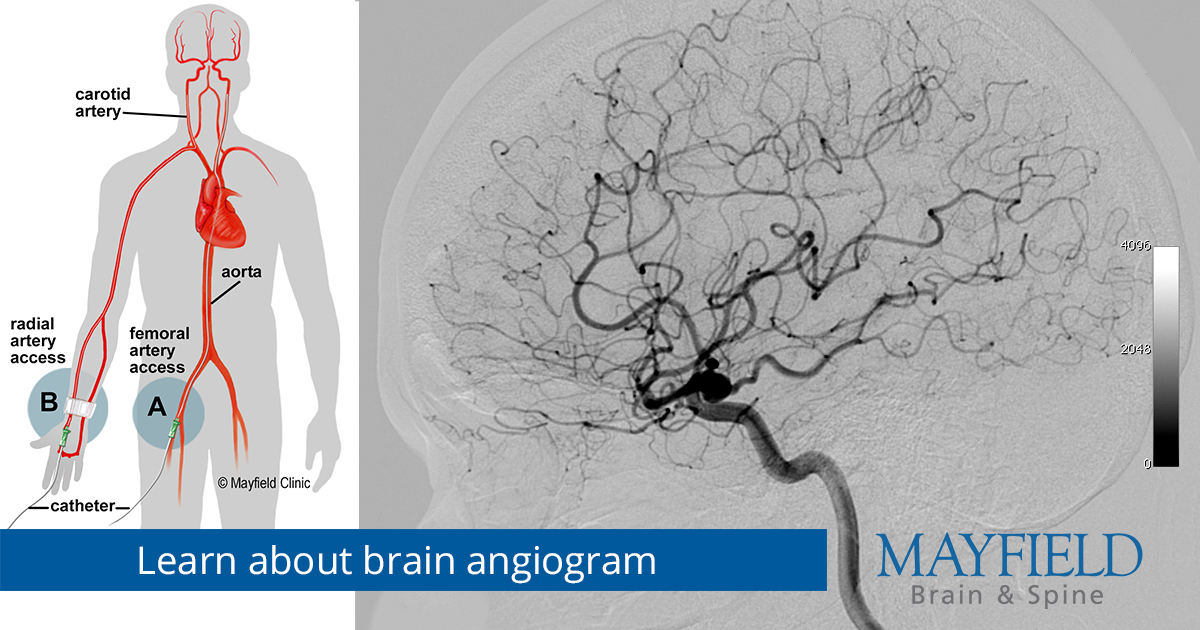
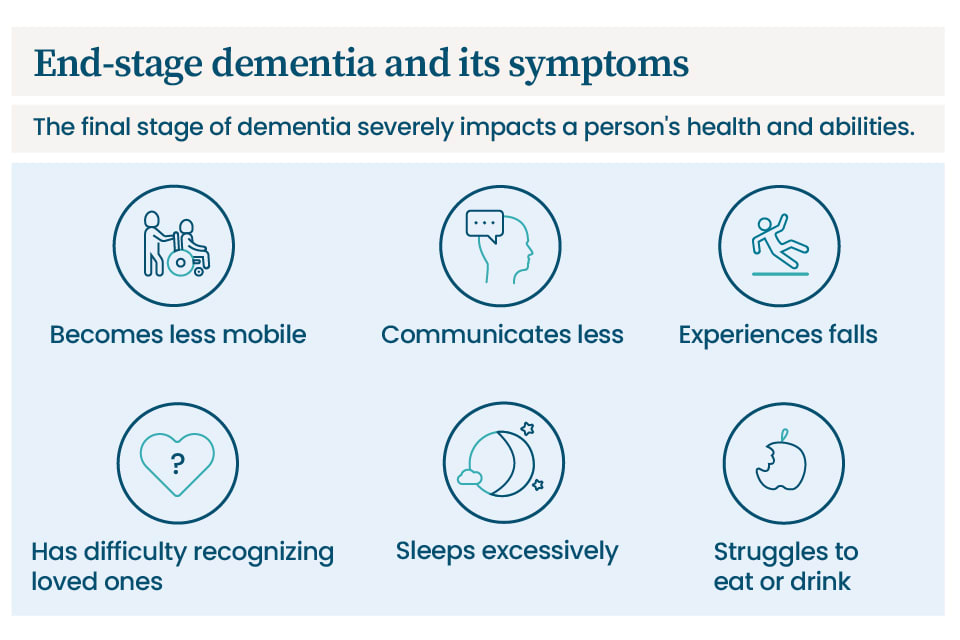
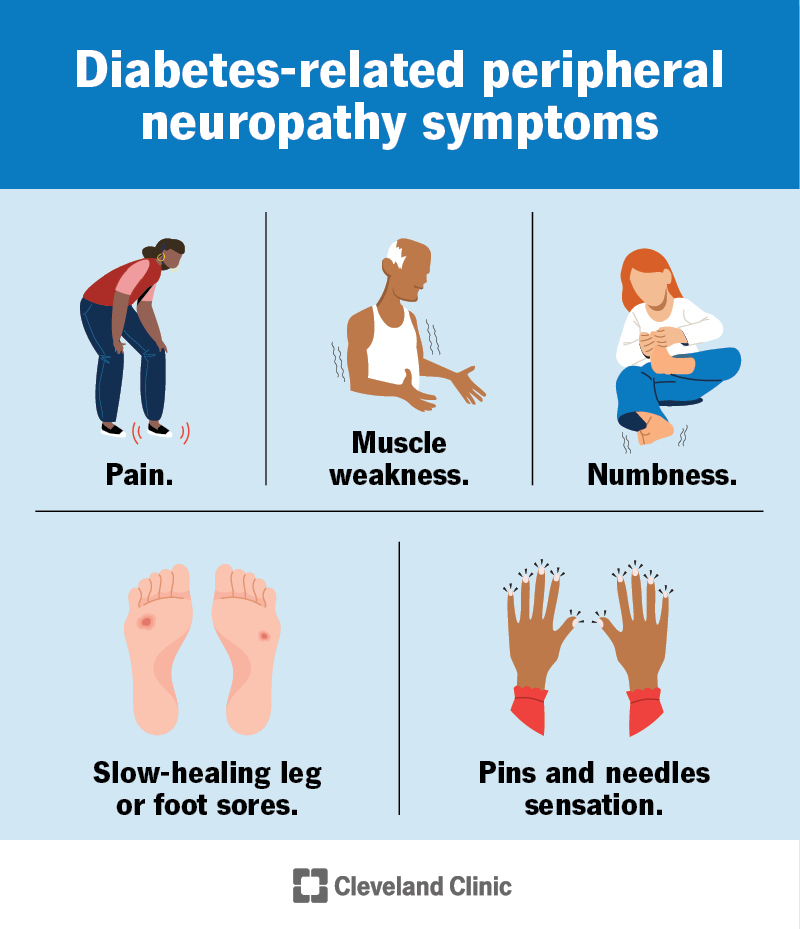
FAQs
What conditions is Epidiolex FDA-approved to treat?
Epidiolex is FDA-approved to treat seizures associated with Lennox-Gastaut syndrome, Dravet syndrome, and tuberous sclerosis complex in patients one year or older.
How is Epidiolex dosed for seizure control?
The typical starting dose is 2.5 mg of CBD per kg of body weight twice daily, gradually increased weekly to a target of 5 mg/kg twice daily. Some patients may require up to 10 mg/kg twice daily, depending on response and tolerance.
What are the common side effects of Epidiolex?
Common side effects include somnolence (sleepiness), decreased appetite, diarrhea, and elevated liver enzymes. These often occur during dose titration and may lessen over time with monitoring.
Can I use over-the-counter CBD products instead of Epidiolex for seizures?
No, Epidiolex is a pharmaceutical-grade, standardized CBD medication with FDA approval and clinical trial support for seizure control. OTC CBD oils vary in quality and dosing and are not recommended for treating seizures.
Is Epidiolex covered by insurance and are there assistance programs?
Many private insurers and Medicare Part D plans cover Epidiolex, often requiring prior authorization. The manufacturer offers copay assistance programs that can reduce out-of-pocket costs by up to 80% for eligible patients.