Short answer: yes, many cancers can actually eat protein, breaking it down into amino acids that fuel their growth, especially when sugar or fat are scarce. But the story is a lot richer than a simple yes-or-no. Understanding how and why cancer cells use protein can clear up a lot of the diet myths youve heard, and it can also point to new treatments that target this sneaky nutrient hacking.
Lets dive in together, keep it friendly, and Ill toss in a few real-world examples so the science feels less like a lecture and more like a chat over coffee.
Cellular Fuel Basics
What do cancer cells feed on?
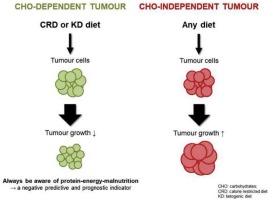
Most of us picture a tumor as a little sweet tooth that guzzles glucose. Thats partly trueglucose is a quick source of energy via glycolysis. However, cancer cells are metabolic shape-shifters. When glucose runs low, they can switch to other fuels: glutamine, fatty acids, and yesextracellular proteins.
How proteins become a fuel source
Proteins in the bloodstream (think albumin, collagen fragments, or even dietary peptides) can be captured by a process called macropinocytosis. The cancer cell essentially gulps down a blob of extracellular fluid, then shuttles the captured proteins to lysosomes. Inside those tiny compartments, enzymes chop the proteins into individual amino acids, which then feed into the TCA cycle or help build new DNA, membranes, and signaling molecules.
Key pathways and players
Research highlighted a protein-feeding pathway named LYSET. When LYSET is active, it streamlines the journey from extracellular protein to usable amino acids. In tumors with mutant KRAS (like many pancreatic cancers), this pathway is supercharged, making protein a significant fuel.
Illustrative example
Imagine a city (the tumor) that usually orders its food (glucose) from a nearby farm. If the farms crops fail, the city starts importing canned goods (proteins) from distant warehouses, opening them up in its own kitchens (lysosomes) to keep the lights on.
Protein vs Sugar vs Fat
Do cancer cells feed on fat?
Yes, they can. Fatty acids can be taken up through transporters like CD36 and then oxidized for energy. Studies show that certain breast and prostate cancers rely heavily on fatty-acid oxidation, especially when glucose is limited.
Do cancer cells feed on sugar?
Glucose remains a primary fuel for many tumors, which is why PET scans use a glucose analogue (FDG) to spot active cancer sites. But sugar isnt the only game in town; its more of a favorite snack than the only food.
Comparing nutrient preferences
| Nutrient | Primary uptake mechanism | Typical tumors that rely on it | Clinical relevance |
|---|---|---|---|
| Glucose | GLUT transporters | Most solid tumors | PETFDG imaging |
| Protein | Macropinocytosis, LRP1 receptors | KRASmutant PDAC, glioblastoma | LYSETtargeted drugs under trial |
| Fatty acids | CD36, FATP | Some breast & prostate cancers | Lipidoxidation inhibitors in development |
This side-by-side view makes it clear: cancer cells are opportunistic eaters, and the nutrient they choose depends on whats most abundant in their microenvironment.
Key Amino Acids
Which amino acids feed cancer cells?
All amino acids can be useful, but a few get a lot of attention:
- Glutamine the classic glutamine addiction many tumors exhibit. It supports nucleotide synthesis and antioxidant defenses.
- Arginine some cancers lack the enzyme to make it, so they hijack external arginine. A study showed that depriving tumors of arginine slows their growth.
- Leucine a potent activator of the mTOR pathway, which drives cell growth.
Essential vs. nonessential amino acids
Essential amino acids (the ones we must get from food) become especially valuable when a tumor's internal synthesis pathways are bottlenecked. Nonessential ones can be made by the cell, but theyre still harvested from the environment when its easier.
Clinical trials targeting amino-acid metabolism
Several early-phase trials are testing drugs that block arginine uptake or glutamine metabolism. While still experimental, the results are promising for tumors that heavily depend on these specific amino acids.
Low-Protein Diets
Can we starve cancer by cutting protein?
Starving sounds dramatic, and its tempting to think that a very low-protein diet could shrink a tumor. The reality is more nuanced. Systemic protein restriction can indeed lower the pool of circulating amino acids, but it also risks muscle wasting, weakened immunity, and poorer tolerance to chemotherapy.
Preclinical evidence
Mouse models where the LYSET pathway was knocked out showed tumors stalling, even when the animals ate normal protein levels. This suggests that the process of protein uptake, rather than the amount of dietary protein, is the therapeutic target.
Human data & safety concerns
A meta-analysis of high-protein diets in cancer patients found no clear link between higher protein intake and faster tumor growth. Conversely, a small pilot trial at Michigan Medicine explored a modest protein-reduction protocol (0.8 g/kg/day) and reported no serious adverse effects, but also no dramatic tumor shrinkage.
Potential downsides
Extreme protein restriction can lead to:
- Loss of lean body mass (sarcopenia)
- Reduced ability to heal from surgery or radiation
- Weaker immune response
Practical guidance for patients
If youre thinking about tweaking your protein intake, start with a conversation. A registered dietitian who specializes in oncology can help you find a sweet spotusually around 0.81.2 g per kilogram of body weight per daybalancing nutrition for recovery with the goal of not feeding the tumor extra fuel. For patients preparing for procedures or recovering after surgery, information about post op recovery can help guide nutritional timing and protein needs during healing.
Whey Protein & Cancer
Does whey protein feed cancer cells?
Whey is a fast-digesting protein rich in leucine. Some lab studies suggest that high leucine levels can stimulate the mTOR pathway, which many cancers hijack for growth. However, human data are far from conclusive. No large-scale trial has shown that consuming whey directly accelerates tumor progression.
What the research says
A 2022 review in Nutrition & Cancer noted that while whey can boost muscle massimportant for patients undergoing chemoit should be used under medical guidance, especially in cancers known to be leucine-sensitive.
Expert perspective
Dr. Ana Lpez, an oncology nutrition specialist, often advises her patients: If you love whey smoothies and they keep you strong, keep thembut tell your team. Well monitor your labs and adjust if needed.
Targeting Protein Pathways
New drugs on the horizon
The most exciting developments focus on shutting down the protein-feeding circuitry itself. LYSET inhibitors are in early-phase trials, aiming to block the lysosomal processing of captured proteins. Another strategy uses macropinocytosis blockers to stop the tumor from gulping extracellular fluid.
Combination strategies
Researchers are testing LYSET inhibitors alongside immunotherapy. The idea is that starving the tumor of protein may make it more visible to the immune system, improving the response to checkpoint inhibitors.
What this means for you
While these therapies are still experimental, they illustrate a shift: instead of blanket diet restrictions, clinicians are developing precision tools that target the tumor's specific metabolic tricks. If youre enrolled in a clinical trial, you may be part of this cutting-edge approach.
Quick Fact Check
Do cancer cells feed on protein? Yes. Many tumors internalize extracellular proteins, break them down in lysosomes, and use the resulting amino acids for energy and building blocks, especially when glucose is scarce.
What amino acids are most important? Arginine, glutamine, and leucine are frequently exploited, but the exact mix varies by cancer type.
Will a high-protein diet boost my tumor? Evidence is mixed. Overall protein intake alone doesnt guarantee faster growth; the tumor's ability to import and process protein matters more.
Is whey protein dangerous for cancer patients? No definitive proof it fuels tumors, but discuss any supplement use with your oncology team to ensure it fits your overall treatment plan.
Can I starve my cancer by cutting protein? Extreme restriction can harm normal tissues. Targeted therapies that block protein-uptake pathways are a safer, research-backed route.
Conclusion
In a nutshell, cancer cells are metabolic chameleons. They can indeed feed on protein, but they also switch between sugar, fat, and amino acids depending on whats available. While the idea of starving a tumor with a low-protein diet sounds appealing, the science tells us that balance, professional guidance, and emerging drugs that block the protein-feeding machinery are far more effective and safer.
If youve ever wondered whether your dinner plate could be feeding a hidden enemy, remember: the bodys nutrition is complex, and so is cancer. Talk to your doctors, consider a personalized nutrition plan, and stay curious. Got a story about how diet impacted your treatment? Share it in the commentsyour experience could help someone else navigate this tricky terrain.
FAQs
Do cancer cells really feed on protein?
Yes, many cancer cells can break down protein into amino acids for energy and growth, especially when glucose or fat are limited.
Which amino acids do cancer cells use most?
Cancer cells often rely on glutamine, arginine, and leucine, which support energy production, DNA synthesis, and cell growth.
Can a low-protein diet starve cancer cells?
Extreme protein restriction can harm healthy tissues and is not proven to shrink tumors; targeted therapies blocking protein uptake are more effective.
Are there drugs that block cancer cells from feeding on protein?
Yes, new drugs targeting pathways like LYSET and macropinocytosis are being tested to stop cancer cells from using protein as fuel.