Short answer: the newest FDAapproved option is tofacitinib (Xeljanz/XeljanzXR), the first oral JAKinhibitor specifically cleared for active ankylosing spondylitis (AS) in 2024. It adds a diseasemodifying choice that works differently from the classic biologic injections.
Why does this matter? Because it gives many patients a convenient pillform alternative, but it also comes with its own set of benefits and risks. Understanding both sides helps you make an informed decision with your rheumatologist and if you're tracking outcomes, consider how ankylosing spondylitis remission is defined and measured in studies and clinics.
Why It Matters
Whats a JAK inhibitor and how does it work?
Think of inflammation as a traffic jam in your immune system. JAK (Janus kinase) proteins are the traffic lights that tell immune cells when to move forward. A JAK inhibitor like tofacitinib flips those lights to stop, reducing the signals that cause the spine and joints to swell.
FDA approval timeline & key studies
The FDA announced the approval in early 2024 after the . In that study, about 44% of participants achieved a 40% improvement in the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), a solid jump compared with placebo.
How this differs from permanent cure myths
Theres no permanent cure for ankylosing spondylitistrustworthy sources like the NHS and Mayo Clinic confirm that all current therapies aim to control inflammation and slow damage. Tofacitinib is a diseasemodifying drug, not a miracle cureitonceandforall pill. Knowing that helps keep expectations realistic and protects you from exaggerated claims you might see online.
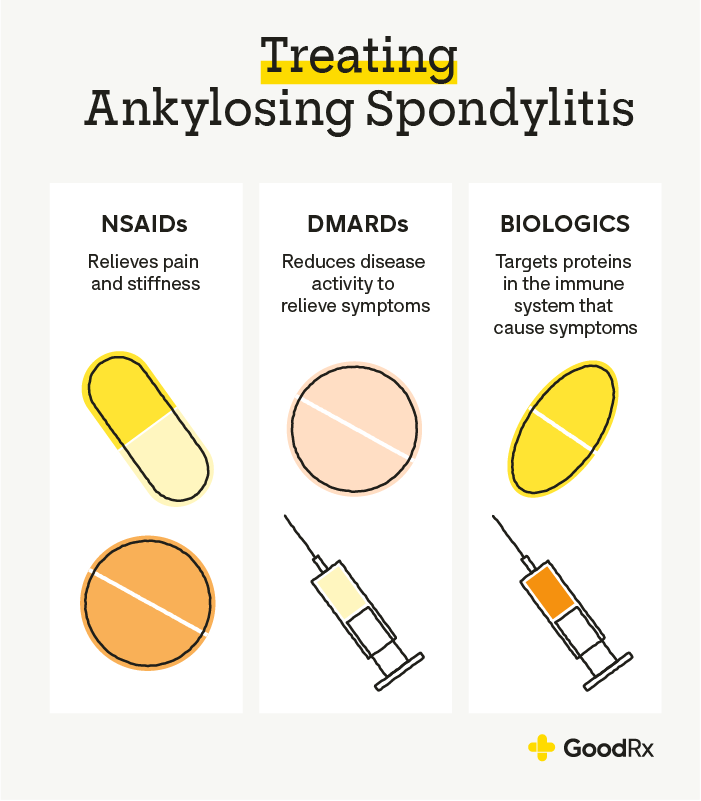
Current Therapy Landscape
Traditional cornerstone: NSAIDs & physiotherapy
Nonsteroidal antiinflammatory drugs (NSAIDs) remain the first line of defence. Theyre cheap, widely available, and can ease morning stiffness for many people. Pair them with a regular physiotherapy routinethink daily stretching, posture drills, and lowimpact cardio. Even with a new JAK inhibitor, doctors still often recommend staying active.
Biologic options still in play
Biologics such as TNF blockers (adalimumab, etanercept) and IL17 inhibitors (secukinumab, ixekizumab) have been the mainstay for over a decade. Theyre administered by injection or infusion and work by neutralising specific inflammatory proteins. If youve already tried a biologic without success, a JAK inhibitor becomes a logical next step.
New entrants beyond JAK inhibitors
Research in 20242025 is buzzing with fresh candidates. Bimekizumab, a dual IL17A/F blocker, showed promising results in early trials, and several IL23/IL12 agents are heading toward PhaseIII. While these are not yet FDAapproved for AS, they illustrate that the pipeline is far from empty.
Injection vs. oral formulations
Oral JAK inhibitors win points for convenienceno clinic visits for needle sticks. However, biologic injections still have a role, especially for patients who cant take oral meds due to gutrelated side effects or who prefer less frequent dosing (some biologics are given once a month).
Who Should Consider
Clinical criteria for eligibility
Generally, youll be a candidate if you have:
- Active disease despite regular NSAID use
- Elevated inflammatory markers (CRP, ESR) or MRI evidence of inflammation
- No uncontrolled infections, recent malignancy, or a history of blood clots
Benefits to expect
Patients on tofacitinib typically report a 4050% drop in pain and stiffness scores within three months. Early imaging data hint at slower radiographic progression, meaning the spine might stay more flexible for longer.
Potential risks & safety monitoring
Like any immunemodulating drug, JAK inhibitors carry warnings for:
- Thromboembolic events (blood clots)
- Serious infections, including opportunistic ones
- Changes in cholesterol and liver enzymes
Doctors usually check a complete blood count, liver function, and lipid panel every 36months. If youve ever had a clot, a JAK inhibitor is probably off the table.
Realworld experience (case vignette)
Take John, a 34yearold graphic designer. After five years on adalimumab with only modest relief, his rheumatologist suggested switching to tofacitinib. Within eight weeks, John could walk his dog again without the usual backtightening pain. He still gets his blood work every four months, and his doctor keeps a close eye on his cholesterol, but the improvement in daily life has been worth the extra monitoring.
Talk With Your Doctor
Key questions to ask
When you sit down with your rheumatologist, consider asking:
- Is a JAK inhibitor the right next step for my disease activity?
- What baseline labs do we need before starting?
- How does the cost compare to my current biologic, and are there patientassistance programs?
Preparing your health data
Bring recent BASDAI scores, any MRI reports, and a list of every medication youre takingincluding overthecounter NSAIDs and supplements. If youve ever had a clot, infection, or cancer, make sure that history is frontandcenter.
Understanding insurance & costsharing
Because tofacitinib now has an FDA label for AS, most insurers will recognize the ICD10 code M45.00. The drugs manufacturer offers a copay assistance card that can dramatically lower outofpocket costs. Your doctors office can usually help you fill out the paperwork.
Shareddecisionmaking tools
Some clinics provide a onepage prosandcons chart that you can take home. You might also find printable PDFs from reputable patienteducation sites like that break down each treatment option in plain language.
Quick Reference Table
| Treatment | Route | FDAApproved Year | Main Target | Typical Efficacy (BASDAI ) | Key Safety Concerns |
|---|---|---|---|---|---|
| Tofacitinib (Xeljanz) | Oral | 2024 | JAKSTAT | 45% | Thrombosis, infections, labs |
| Upadacitinib (Rinvoq) | Oral | 2022 | JAKSTAT | 40% | Same as above |
| Secukinumab (Cosentyx) | SC injection | 2016 | IL17A | 38% | Candida infections |
| Adalimumab (Humira) | SC injection | 2006 | TNF | 35% | TB reactivation |
| Bimekizumab (investigational) | SC injection | IL17A/F | 4050% (PhaseIII) | Oral/genital candidiasis |
Bottom Line
The newest FDAapproved treatment for ankylosing spondylitis is an oral JAK inhibitorspecifically tofacitinib. It offers a convenient, diseasemodifying option that can dramatically improve pain and stiffness for many patients, yet it is not a permanent cure. Safety monitoring, personal risk factors, and cost considerations all play a role in the decision.
Ultimately, the best medicine for ankylosing spondylitis is the one that fits your lifestyle, disease activity, and health profile. Talk openly with your rheumatologist, bring your questions, and weigh the pros and cons together. If youve tried other therapieswhether NSAIDs, biologic injections, or even experimental trialsthis new age of oral treatments might finally feel like the fresh start youve been waiting for.
Whats your experience with the newer AS therapies? Have you started a JAK inhibitor, or are you still on an older regimen? Share your story in the commentsyour insight could help someone else navigate this complex journey.
If you have any lingering questions or need help preparing for your next appointment, feel free to reach out. Were in this together, and every step toward better control of ankylosing spondylitis is a win.