Hey there! If youve just heard the name VYONDYS53 and feel a mix of hope and confusion, youre not alone. In a nutshell, VYONDYS53 is a specialized therapy that helps a slice of Duchenne muscular dystrophy (DMD) patients make a tiny but crucial change in their DNA, letting their bodies produce a shortened, functional version of the missing protein.
Heres the quick scoop youve been looking for: the drug received , its also cleared in Europe by the EMA, costs roughly $476,000 per year in the U.S., and it works by skipping exon53 during gene transcription. Below well unpack every detailbenefits, risks, price, and what to expectso you can feel confident talking to a doctor or a loved one.
Approval Timeline
When did VYONDYS53 get FDA approval?
The United States Food and Drug Administration gave the green light on August192019. It was a landmark decision because it was the first antisenseoligonucleotide therapy approved for a DMD mutation that isnt a nonsense mutation.
What about European approval?
The European Medicines Agency (EMA) followed a couple of years later, officially authorizing VYONDYS53 for use across the EU in 2021. This means families in France, Germany, Spain, and other member states can now access the treatment under their national health systems.
How does the FDA label define its use?
The states that VYONDYS53 is indicated for DMD patients four years of age or older who have a confirmed mutation amenable to exon53 skipping. The label also spells out dosing (30mg/kg weekly IV infusion) and the required safety monitoring.
Is the drug available outside the U.S. and Europe?
Aside from the U.S. and EU, Sarepta Therapeutics (the maker) has pursued compassionateuse agreements in Canada, Japan, and a few MiddleEastern countries. Availability varies, but many families can apply through local regulatory pathways or special access programs.
How It Works
What is exonskipping, and why exon53?
Imagine your DNA as a long paragraph that accidentally includes a typoan extra word that throws off the entire sentence. In many DMD cases, a missing piece of the dystrophin gene (exon53) creates a frameshift typo, resulting in a broken protein. VYONDYS53 acts like a precise editor, telling the cells machinery to skip that typo, restoring the reading frame and producing a shorter but still functional dystrophin protein.
How does golodirsen achieve the skip?
Golodirsen is a phosphorodiamidate morpholino oligomer (PMO). Its a short, synthetic strand that binds specifically to the premessenger RNA at the exon53 splice site. By masking this region, the splicing engine simply jumps over exon53, stitching the surrounding exons together.
Visual analogy
Think of a movie reel with a damaged frame. Instead of trying to repair the frame, you cut it out and splice the film so the story still plays smoothly. Thats essentially what golodirsen does at the molecular level.
What evidence backs this mechanism?
The pivotal PhaseII trial (Sareptas NCT02386998) showed a modest increase in dystrophin levelsabout 0.51% of normalin muscle biopsies after 48 weeks. While that sounds tiny, clinical endpoints hinted at a slower decline in the sixminute walk test and North Star ambulatory scores compared with historical controls.
Who Benefits
Which patients are eligible?
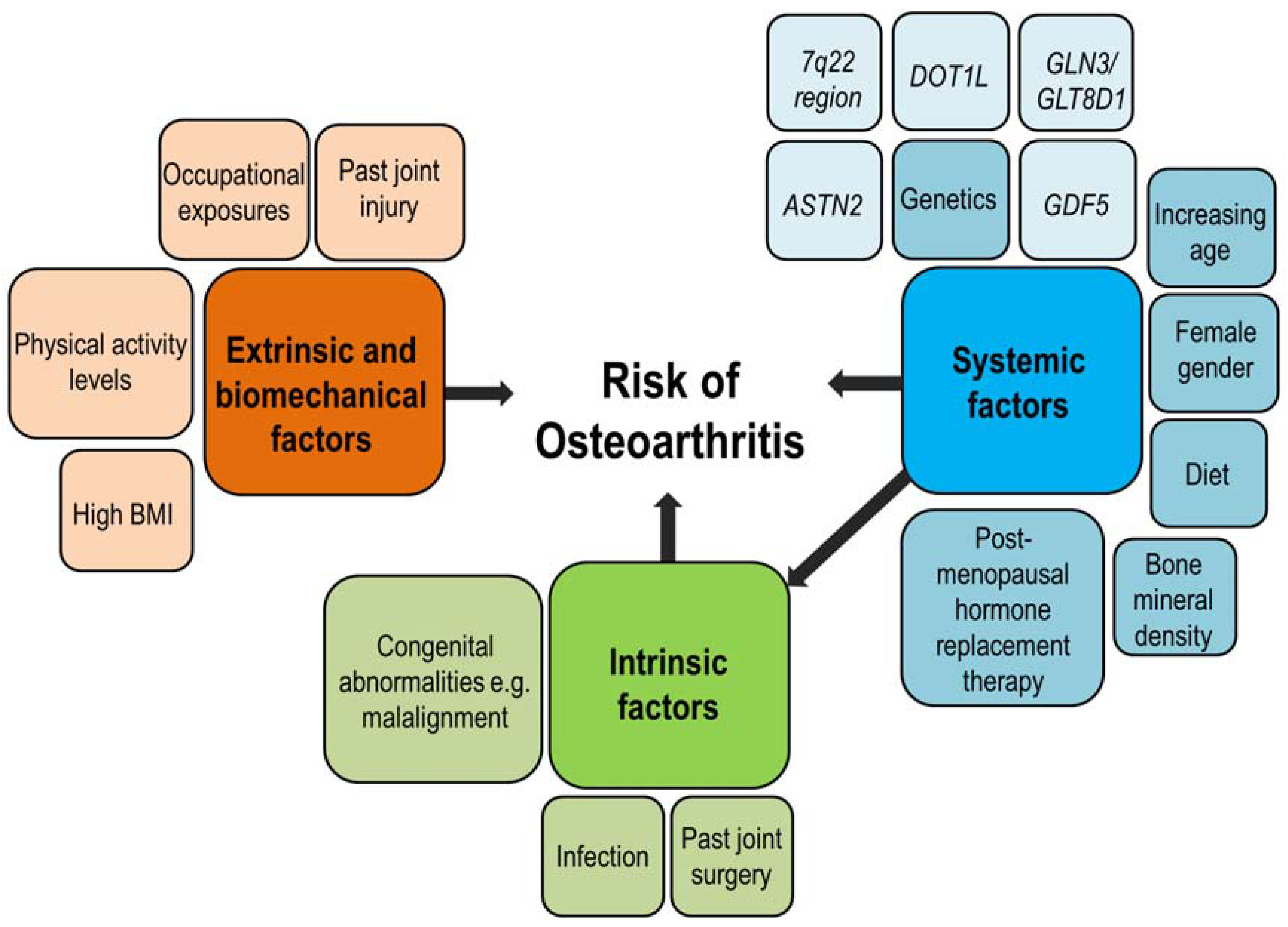
Eligibility hinges on two things: a confirmed exon53skippable mutation (roughly 810% of all DMD cases) and age 4years. Genetic testing by a certified laboratory is the first stepif the test shows a deletion or duplication that removes exon53, youve got a candidate.
Why the age requirement?
Earlystage disease offers the best chance for any therapy to preserve muscle function. Starting before significant fibrosis sets in maximizes the potential benefit of the modest dystrophin boost.
Realworld story
Take Alex, a 6yearold from Ohio. His family discovered his DMD mutation during a routine genetic panel. After a candid conversation with their neurologist, they started VYONDYS53. Six months in, Alexs parents reported that his energy levels felt steadier, and he could keep up with his classmates a bit longer than expected. Its not a miracle cure, but it gave them a tangible reason to hope.
Dosing & Safety
Whats the dosing schedule?
The label recommends a 30mg/kg weekly intravenous infusion. Most infusion centers administer it over 3045 minutes, monitoring vitals before, during, and after the session.
Common sideeffects?
Patients may experience mild infusionrelated reactionsflushing, itching, or a lowgrade fever. Renal labs are checked at baseline and then every three months, as the drug is cleared through the kidneys. Serious adverse events are rare, but clinicians stay vigilant for signs of hypersensitivity.
Who should avoid VYONDYS53?
Severe renal impairment, known hypersensitivity to morpholino oligomers, or a history of severe infusion reactions are red flags. In those cases, alternative therapies or clinical trial enrollment may be a better fit.
Safety checklist for you and your doctor
| Step | What to Do |
|---|---|
| Baseline labs | Kidney function (eGFR), CBC, liver panel |
| Preinfusion vitals | BP, heart rate, temperature |
| During infusion | Observe for rash, flushing, breathing changes |
| Postinfusion | Monitor vitals for 30min, repeat labs q3months |
Cost & Access
How much does VYONDYS53 cost?
The list price in the United States hovers around $476,000 per patient per year. That number can feel overwhelming, but it reflects the complexity of manufacturing antisenseoligonucleotides and the limited patient pool.
Insurance and assistance options
Most large insurers will negotiate coverage, especially if you have a genetic confirmation. Sarepta offers a copay assistance program that can reduce outofpocket costs dramatically for eligible families. Additionally, Medicaid in many states lists VYONDYS53 as a covered specialty drug.
How does the price compare internationally?
European pricing is generally lower, thanks to national healthtechnology assessments. For example, Germanys negotiated price is roughly 300,000 per year, while the UKs NHS has secured a confidential discount that brings the annual cost down to about 250,000.
Price comparison
| Region | Annual List Price |
|---|---|
| United States | $476,000 |
| European Union (average) | 300,000 |
| Canada | CAD350,000 |
What can you do now?
Start by gathering your genetic report and reaching out to a DMDspecialized neurologist. Ask about the assistance program and whether your insurer needs prior authorization. Remember, the paperwork can be a marathon, but every step brings you closer to access.
Sales & Market
How are sales trending?
Sarepta reported global revenues of about $1.4billion in 2023, with VYONDYS53 accounting for roughly 15% of that figure. While sales have plateaued in the U.S. due to the niche patient population, the drug continues to generate steady income as more patients become eligible.
Future outlook?
With nextgeneration exonskipping candidates and emerging genetherapy approaches, VYONDYS53 may eventually share the stage with even more powerful treatments. Yet, its current niche still offers a valuable option for families who arent candidates for gene therapy or who need a bridge while awaiting other trials.
Key Resources
For deeper dives, check out these trusted sources:
Conclusion
VYONDYS53 isnt a magic bullet, but its a scientifically sophisticated option that has opened doors for a subset of DMD patients who previously had no targeted therapy. By understanding the approval history, how the drug works, who can benefit, and what the price tag looks like, youre better equipped to have an informed conversation with your healthcare team. If you think you or a loved one might qualify, grab the , reach out to a specialist, and explore the assistance programs that could ease the financial burden.
Wed love to hear your thoughtshave you spoken with a doctor about VYONDYS53? What questions are still on your mind? Drop a comment below or reach out; together we can navigate this complex landscape.
FAQs
What is VYONDYS 53 and who is it for?
VYONDYS 53 (golodirsen) is an FDA-approved antisense oligonucleotide therapy for Duchenne muscular dystrophy (DMD) patients with a confirmed genetic mutation amenable to exon 53 skipping, typically affecting about 8% of DMD cases[1][4][7].
How does VYONDYS 53 work?
VYONDYS 53 works by “skipping” exon 53 during gene transcription, allowing the body to produce a shorter but still functional version of the dystrophin protein, which is otherwise missing or defective in DMD[7][9].
What are the benefits and clinical results of VYONDYS 53?
Clinical trials showed an increase in dystrophin levels (from less than 0.1% to just over 1% of normal) after 48 weeks, and some data suggest a slower decline in walking and lung function compared to untreated patients, though the clinical benefit is still being confirmed in ongoing studies[3][7].
What are the common side effects of VYONDYS 53?
The most common side effects include headache and fever. Most reactions are mild, but kidney function monitoring is required, and the drug is not recommended for those with severe kidney impairment or hypersensitivity to similar drugs[6].
How much does VYONDYS 53 cost and how can patients access it?
In the U.S., VYONDYS 53 costs approximately $476,000 per year. Insurance coverage varies, and Sarepta offers assistance programs. Availability and pricing differ internationally, with European national health systems often providing access at lower prices[22].