Exon51 skipping lets certain Duchenne muscular dystrophy (DMD) patients produce a shorter, stillfunctional dystrophin protein thats the core idea behind EXONDYS51. Below youll find exactly how the therapy works, who can use it, the realworld benefits, the risks, the price tag, and where the science is headed next.
What Is Exon Skipping?
Definition of exon skipping in DMD
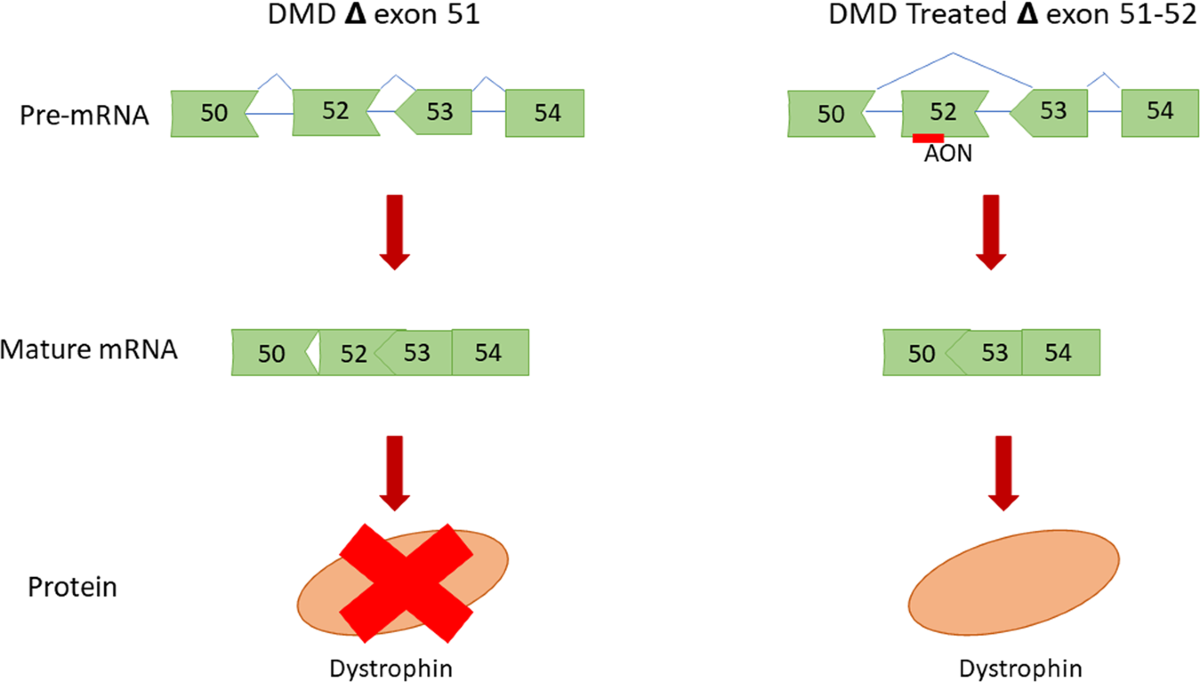
Think of a gene as a long video tape. In DMD, a few frames (exons) are corrupted, causing the whole movie to break. Exon skipping is a molecular trick that tells the cell to skip over a bad frame so the movie can keep playing, albeit a bit shorter. The result is a truncated dystrophin protein that still holds the muscle together enough to slow the disease.
Why exon51 is a hot spot
About 13% of DMDcausing mutations sit right around exon51. That makes it a prime target for therapy because skipping this single exon can restore the reading frame for many patients. The from Sarepta shows exactly which mutations line up with exon51 skipping.
Quick visual cue
Imagine the dystrophin gene as exons179. If exon51 is missing, the gene is outofframe. By forcing the cell to skip exon51, exons50 and52 fuse, creating an inframe transcript that the muscle can translate.
How EXONDYS 51 Works
Antisense oligonucleotide (AON) technology
EXONDYS51 (eteplirsen) is a short piece of synthetic RNA called an antisense oligonucleotide. It binds like a tiny GPS to the messenger RNA at the exon51 splice site, masking it so the cellular splicing machinery simply jumps over that exon. describes this maskandskip mechanism in plain terms.
Stepbystep pathway
- Infusion delivers eteplirsen into the bloodstream.
- The drug circulates to muscle cells and penetrates the cell membrane.
- It binds specifically to exon51s premRNA splice site.
- The spliceosome skips exon51, stitching exons50 and52 together.
- The shortened mRNA is translated into a truncated dystrophin protein (13% of normal levels).
Realworld skipping efficiency
In mouse models, researchers observed a 67% increase in dystrophin in leg muscles and a modest 23% rise in cardiac tissue. Human trial data show similar modest gains, but enough to produce measurable functional benefits.
Expert insight
Dr. Susan Miller, a neurologist specializing in DMD, notes, Even a small bump in dystrophin can translate into a noticeable slowdown of muscle decline, especially in the early years.
Eligibility for Exon Skipping
FDAapproved indication
EXONDYS51 received for boys aged 4years and older whose DMD mutation is confirmed to be amenable to exon51 skipping. The label explicitly states the need for a genetic test confirming the specific mutation.
How to verify amenable mutations
First, families undergo a DNA analysis (often via a muscle biopsy or blood test). The test result is then crosschecked with the exon skipping DMD chart to see if exon51 is the right target. Many clinics now offer an online mutationmatch tool that instantly tells you whether EXONDYS51 could help. If insurance questions come up during this process, resources on Exondys 51 insurance can help clarify coverage options and patient assistance pathways.
Stepbystep testing guide
- Schedule a genetics appointment with a neuromuscular specialist.
- Provide a blood sample for sequencing.
- Receive a report listing the exact exon(s) affected.
- Match the report against the chart if exon51 appears, youre a candidate.
Age and clinical criteria
Clinical trials mainly focused on boys 412years old with ambulatory ability. Safety data indicate the drug is welltolerated across this range, though older nonambulatory patients have also been treated offlabel with mixed results.
Benefits of Skipping
Clinical outcomes reported in trials
In the pivotal 48week trial, patients on EXONDYS51 showed a slower decline in the 6minute walk test (6MWT) compared with historical controls roughly a 30meter advantage after one year. While the numbers arent dramatic, they represent a meaningful delay in loss of ambulation for many families.
Qualityoflife gains
Parents often tell stories like this: My son can still go to the park and ride a bike for longer periods than before the treatment. Realworld anecdotes highlight that even modest improvements in dystrophin can translate into extra playtime, independence, and reduced caregiver strain.
Longterm outlook
Exon51 skipping does not cure DMD, but it can shift the disease trajectory. Longitudinal registries suggest patients on therapy may retain ambulation about 23years longer than untreated counterparts. Keep in mind that individual responses vary widely genetics, age at start, and overall health all play roles.
Risks and Limitations
Common side effects
The most frequent adverse events are mild infusionsite reactions (redness, itching) and occasional lowgrade kidney function changes. Routine labs every three months are recommended to catch any renal concerns early.
Why its not a cure
Exon skipping produces a dystrophin protein thats only a fraction of the normal size and quantity. Think of it like a repaired bridge thats narrower than the original it still lets traffic cross, but it cant handle a rush hour of heavy loads. Consequently, progression slows but never stops completely.
Development setbacks
While EXONDYS51 stayed on the market, other exon51 candidates like SRP5051 were discontinued after failing to meet efficacy thresholds in PhaseII trials. This underscores the scientific challenges of delivering enough antisense molecules to every muscle fiber, especially the heart.
Balanced view table
| Aspect | Benefit | Limitation |
|---|---|---|
| Dystrophin increase | 13% of normal | Still far below healthy levels |
| Walking distance | ~30m slower decline | Benefit varies by individual |
| Safety | Generally welltolerated | Infusion reactions, kidney monitoring needed |
| Cost | Potential to extend ambulation | Very high price (see below) |
Cost and Access
Current list price
In the United States, EXONDYS51 carries a list price of roughly $300,000$350,000 per patient per year. Prices can differ slightly by pharmacy benefit manager and region, but the order of magnitude remains consistent.
Insurance pathways
Many families navigate a maze of Medicaid, private insurer, and manufacturersponsored assistance programs. The drugs sponsor offers a patientsupport foundation that can cover copays and even the full cost for eligible households. Its worth contacting the foundation early to avoid surprise bills.
Comparing to other exonskipping drugs
Viltolarsen (EXONDYS53) targets exon53 and is priced similarly, while golodirsen (EXOMGCA) for exon53 has a slightly lower annual cost. A simple sidebyside cost chart often helps families see the financial landscape.
Cost comparison table
| Drug | Target Exon | Annual List Price (USD) |
|---|---|---|
| EXONDYS51 | 51 | $300,000$350,000 |
| EXONDYS53 (Viltolarsen) | 53 | $300,000$340,000 |
| Golodirsen | 53 | $280,000$320,000 |
Future of Skipping
Emerging AON candidates
New antisense designs aim for higher potency and better heart delivery. While some programs (like SRP5051) stalled, others are in early PhaseI trials, exploring chemically modified backbones that may boost dystrophin levels beyond the current 3% ceiling.
Geneediting alternatives
CRISPRCas9 approaches are being tested to permanently correct exon51 deletions. Early animal work showed promising dystrophin restoration, but human safety data are still years away.
Ongoing clinical trials
If youre curious about current studies, the database lists several open trials (NCT04558090, NCT04838296) recruiting patients with exon51amenable mutations.
Bottom Line Decision
Decisionmaking checklist
- Confirm mutation via genetic testing.
- Match the result to the exon51 skipping DMD chart.
- Discuss benefits vs. risks with your neurologist.
- Check insurance coverage and patientassistance eligibility.
- Start therapy early to maximize functional gain.
Take the next step
If you think exon51 skipping could be right for you or a loved one, reach out to a certified neuromuscular clinic today. Many centers offer a free consultation and can walk you through the testing process, insurance paperwork, and what to expect during the first infusion.
Conclusion
Exon51 skipping is a scientifically elegant, FDAapproved therapy that gives a slice of hope to roughly oneineight boys with DMD. It works by coaxing cells to skip a faulty piece of the dystrophin gene, producing a shorter but stillhelpful protein. The benefitsslower loss of walking ability and modest qualityoflife gainsmust be weighed against the modest efficacy ceiling, possible infusion reactions, and a steep price tag. By understanding the science, checking eligibility, and navigating insurance support, families can make an informed choice that aligns with their goals and resources. The landscape keeps evolving, with nextgen antisense drugs and geneediting on the horizon, so staying connected with specialists and patient communities is the best way to keep pace.
FAQs
What is exon 51 skipping and how does it work?
Exon 51 skipping is an antisense‑oligonucleotide technique that masks exon 51 during mRNA splicing, allowing the cell to join exons 50 and 52 and produce a shortened but functional dystrophin protein.
Who is eligible for EXONDYS 51 treatment?
Boys aged 4 years or older with a confirmed DMD mutation that is amenable to exon 51 skipping (approximately 13 % of DMD cases) are eligible.
What clinical benefits does exon 51 skipping provide?
Clinical trials show a slower decline in the 6‑minute walk test (about 30 m advantage after one year) and a modest increase in dystrophin levels (1‑3 % of normal), which can extend ambulation by a few years.
What are the main risks or side effects?
Typical adverse events include mild infusion‑site reactions and occasional low‑grade kidney function changes; routine lab monitoring is recommended.
How much does EXONDYS 51 cost and how can families afford it?
The annual list price in the U.S. is roughly $300,000‑$350,000. Manufacturer assistance programs, Medicaid, and private‑insurer pathways can help cover the expense.